Course
Ohio APRN Renewal Bundle – Part 1
Course Highlights
- In this course on safely prescribing opioids, you’ll be able to describe factors when prescribing opioids, their indications, and their effects.
- You’ll also be able to explain new CDC guidelines for opioid prescribing.
- you’ll also be able to identify appropriate teaching elements for patients when prescribed opioids.
About
Total Contact Hours Awarded: 24 , including 12 pharmacological hours
Course By:
Multiple Authors
Begin Now
Read Course | Complete Survey | Claim Credit
➀ Read and Learn
The following course content
Safe Prescribing of Opioid Drugs
Introduction
For centuries, humans have been using, developing, and synthesizing opioid compounds for pain relief. Opioids are essential for treating patients who are experiencing severe and sometimes even moderate pain. Chronic pain can negatively affect our lives. In 2011, the cost of chronic pain ranged from $560 to $635 billion in direct medical expenses, lost productivity, and disability. An estimated one in five U.S. adults had chronic pain (11).
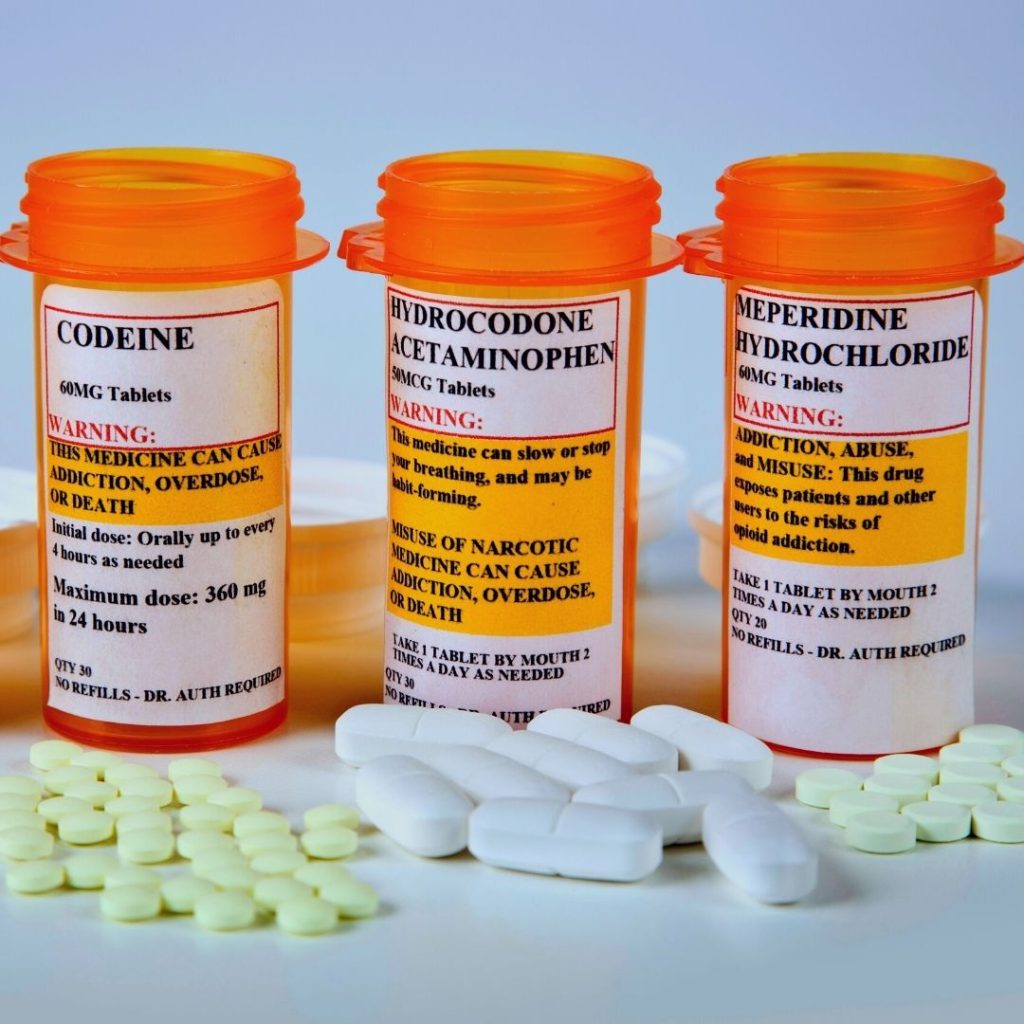
Introduction of opioid drug class, indications for use, most prescribed opioids, and their effects.
Opioids usually bind to mu-opioid receptor sites, where they have agonist effects, providing pain relief, sedation, and sometimes feelings of euphoria. Opiates refer only to natural opioids derived from the poppy plant. The term "opioids" includes all-natural, semi-synthetic, and synthetic opioids.
Opioids are classified into several categories based on their origin and chemical structure:
- Natural Opioids (Opiates): These come from the opium poppy plant. Examples include morphine and codeine.
- Semi-Synthetic Opioids: These are natural opioids that are chemically modified. Examples include drugs like oxycodone, hydrocodone, oxymorphone, and hydromorphone.
- Synthetic Opioids: These opioids are entirely synthesized in a laboratory and do not have a natural source. Examples include fentanyl, tramadol, and methadone (4).
Providers must always caution patients about the benefits and risks. The main advantage of opioid medication is that it will reduce pain and improve physical function. A provider may use a 3-item Pain, Enjoyment of Life, and General Activity (PEG) Assessment Scale:
- What number best describes your pain on average in the past week?
- What number best describes how, during the past week, pain has interfered with your enjoyment of life?
- What number best describes how, during the past week, pain has interfered with your general activity?
The desired goal is a 30% improvement overall with opioid treatment. (Centers for Disease and Control)
Providers must also go over potential side effects and warnings. Side effects include sedation, dizziness, confusion, nausea, vomiting, constipation, itching, pupillary constriction, and respiratory depression. Providers should discuss the importance of taking medication as prescribed. Taking opioids in larger than prescribed dosage, or in addition to alcohol, other illicit substances, or prescription drugs, can lead to severe respiratory depression and death. Individuals should not drive when taking opioids due to the sedating effects and decreased reaction times.
There are now thousands of different Food and Drug Administration (FDA) approved opioids available for providers to prescribe. They can be administered via other routes and come in various potencies. The strength of morphine is the "gold standard" used when comparing opioids. A morphine milligram equivalent (MME) is the degree of µ-receptor agonist activity. The following is a sample of some of the most prescribed opioids and their MME:
| Opioid | Conversion factor | Opioid | Conversion factor |
| Codeine | 0.15 | Morphine | 1.0 |
| Fentanyl transdermal (in mcg/hr) | 2.4 | Oxycodone | 1.5 |
| Hydrocodone | 1.0 | Oxymorphone | 3.0 |
| Hydromorphone | 5.0 | Tapentadol† | 0.4 |
| Methadone | 4.7 | Tramadol§ | 0.2 |
To utilize this information, multiply the dose for each opioid by the conversion factor to determine the quantity in MMEs. For example, tablets containing hydrocodone 5 mg and acetaminophen 325 mg taken four times a day would include a total of 20 mg of hydrocodone daily, equivalent to 20 MME (11).

Self Quiz
Ask yourself...
- How often do you prescribe opioids to your patients? If so, which ones and what dosages?
- If you do not prescribe opioids, do you treat patients taking them?
- Have you ever felt hesitant about prescribing opioids? What were the reasons?
- What topics do you routinely cover when you discuss opioid prescriptions with your patients?
- Do you know how to determine if a patient is engaging in opioid-seeking behaviors?
A history of opioids leading to the current epidemic
While the current opioid epidemic has caused devastating effects in recent years, destruction from opioids has been going on for centuries. In the early 1800s, physicians and scientists became aware of the addictive qualities of opium. This finding encouraged research to develop safer ways to deliver opioids for pain relief and cough suppression, which led to the development of morphine. By the mid-1800s, with commercial production and the invention of the hypodermic needle, morphine became easier to administer.
During the Civil War (1861 to 1865), injured soldiers were sometimes treated with morphine, and some developed lifelong addictions after the war. Without other options for pain relief, physicians kept giving patients morphine as treatment. Early indicators that morphine should be used cautiously were largely ignored. Between 1870 and 1880, the use of morphine tripled. Even with the problems associated with opioids, they continued to serve a vital part in the pain treatment of patients, and their use and development continued (6).
Opioid prescribing increased fourfold during 1999–2010. Along with the increase in opioid prescriptions during this time, how they were prescribed also changed; opioids were increasingly prescribed at higher dosages and for longer durations. The number of people who reported using OxyContin for non-medical purposes increased from 400,000 in 1999 to 1.9 million in 2002 and to 2.8 million in 2003. This was accompanied by an approximately fourfold increase in overdose deaths involving prescription opioids (8).
In 2020, approximately 1.4 million people were diagnosed with opioid use disorder (OUD), of those associated with opioid painkillers, as opposed to 438,000 who have heroin-related OUD (1, 4). Over 100,000 people died of a drug overdose, with 85% involved in an opioid (1, 4).
Widespread efforts were made to combat this growing issue. The prescribing rate peaked and leveled off from 2010-2012 and has been declining since 2012. In 2021, an estimated 2.5 million adults had been diagnosed with OUD. However, the amount of MME of opioids prescribed per person is still around three times higher than in 1999 (5).
Controlled Substances
On July 1, 1973, the Drug Enforcement Administration (DEA) was established in the United States. The Diversion Control Division oversees pharmaceuticals. Within this Division are five levels of controlled substances, which classify illicit and medicinal drugs (7).
Schedule I Controlled Substances
No medical use, lack of accepted safety for use under medical supervision, and high abuse potential.
Examples of Schedule I substances are heroin, lysergic acid diethylamide (LSD), and marijuana (cannabis).
Schedule II/IIN Controlled Substances (2/2N)
High potential for abuse, which can lead to severe dependence.
Examples include hydromorphone, methadone, meperidine, oxycodone, and fentanyl. Other narcotics in this class include morphine, opium, codeine, and hydrocodone.
Some examples of Schedule IIN stimulants include amphetamine, methamphetamine, and methylphenidate.
Other substances include amobarbital, glutethimide, and pentobarbital.
Schedule III/IIIN Controlled Substances (3/3N)
Less potential for abuse than substances in Schedules I or II, and abuse may lead to moderate or low physical dependence or high psychological dependence.
Include drugs containing not more than 90 milligrams of codeine per dosage unit like Acetaminophen with Codeine and buprenorphine.
Schedule IN non-narcotics includes benzphetamine, phendimetrazine, ketamine, and anabolic steroids such as Depo®-Testosterone.
Schedule IV Controlled Substances
Have a low potential for abuse relative to substances in Schedule III.
Examples include alprazolam, carisoprodol, clonazepam, clorazepate, diazepam, lorazepam, midazolam, temazepam, and triazolam, Tramadol.
Schedule V Controlled Substances
Low potential for abuse relative to Schedule IV and primarily consist of medications that have small quantities of narcotics.
Examples include cough preparations containing not more than 200 milligrams of codeine per 100 milliliters or per 100 grams and ezogabine (10).

Self Quiz
Ask yourself...
- How have your prescribing practices of opioids changed in response to the epidemic over time?
- What trends have you noticed in overall inpatient treatment plans regarding opioid prescribing trends (e.g., dose changes? or increase in opioid alternative therapies?)
- Do you think there is a stigma surrounding patients who are currently using opioids?
Explore behaviors that indicate opioid seeking, misuse, or addiction in patients.
Opioid use disorder (OUD) causes significant impairment or distress. Diagnosis is based on the following criteria: unsuccessful efforts to reduce or control use or use that leads to social problems and a failure to fulfill obligations at work, school, or home. The term Opioid Use Disorder is the preferred term; "opioid abuse or dependence" or "opioid addiction" have negative connotations and should be avoided. (3).
OUD occurs after a person has developed tolerance and dependence, resulting in a physical challenge to stop opioid use and increasing the risk of withdrawal. Tolerance happens over time when a person experiences a reduced response to medication, requiring a larger amount to experience the same effect. Opioid dependence occurs when the body adjusts to regular opioid use. Unpleasant physical symptoms of withdrawal occur when medication is stopped. Symptoms of withdrawal include anxiety, insomnia, abdominal pain, vomiting, diarrhea, etc. (5).
Patients who have OUD may or may not have practiced drug misuse. Drug misuse, the preferred term for "substance abuse," is the use of illegal drugs and the use of prescribed drugs other than as directed by a doctor, such as using more amounts, more often, or longer than recommended or using someone else's prescription (3).
Some indications that a patient may be starting to have unintended consequences with their opioid prescription may include the following symptoms: craving, wanting to take opioids in higher quantities or more frequently, difficulty controlling use, or work, social, or family issues. If providers suspect OUD, they should discuss their concerns with their patients nonjudgmentally and allow the patient to disclose related concerns or issues. Providers should assess the presence of OUD using the DSM-5 criteria.
Providers can use validated screening tools such as:
- Urine and oral fluid toxicology testing
- Drug Abuse Screening Test (DAST)
- Tobacco, Alcohol, and/or other Substance use Tools (TAPS)
- A three-question version of the Alcohol Use Disorders Identification Test (AUDIT-C)
The following patients are at higher risk for OUD or overdose:
- History of depression or other mental health conditions
- History of a substance use disorder
- History of overdose
- Taking 50 or greater MME/day or taking other central nervous system depressants with opioids


Self Quiz
Ask yourself…
- How often do you have patients exhibiting symptoms of OUD in your practice setting?
- What would your next steps be if you identify a patient with potential OUD (e.g., additional screening, referral, or treatment plans)?
- Have you noticed any trends in patients presenting with OUD? Such as socioeconomic status, occupation, gender, race, and medical diagnosis.
The 12 components of the CDC’s recent guidelines for opioid prescribing.
In 2022, the CDC updated the 2016 guidelines to help prescribers navigate prescribing opioids amid an epidemic. These guidelines are directed toward prescribing medications to adults to be taken in the outpatient setting, for example, primary care clinics, surgery centers, urgent cares, and dental offices. These do not apply to providers caring for individuals with sickle-cell disease, cancer, those receiving inpatient care, or end-of-life or palliative care. They are also intended to serve as a guideline, and each treatment plan should be specific to the unique patient and circumstances.
Some of the Goals of the guidelines are to:
- Improve communication between providers and patients about treatment options and discuss the benefits and risks before initiating opioid therapy.
- Improve the effectiveness and safety of treatment to improve quality of life.
- Reduce risks associated with opioid treatment, including opioid use disorder (OUD), overdose, and death.
Recommendation #1: Determining when it is appropriate to initiate opioids for pain.
An essential part of the prescribing process is determining the anticipated pain severity and duration based on the patient’s diagnosis. Pain severity can be classified into three categories when measured using the standard 1-10 numeric scale. Pain scores 1-4 are considered mild, 5-6 are moderate, and 7-10 severe. Opioids are typically used for moderate to severe pain.
The patient’s diagnosis will allow the provider to determine if pain initially falls into one of the following three categories of anticipated duration: acute, subacute, and chronic. Acute pain is expected to last for one month or less. Acute pain often is caused by injury, trauma, or medical treatments such as surgery. Unresolved acute pain may develop into subacute pain if not resolved in 1 month. If pain exceeds three months, it is classified as chronic. Pain persisting longer than three months is chronic. It can result from underlying medical conditions, injury, medical treatment, inflammation, or unknown cause.
The CDC guidelines state that non-pharmacologic and non-invasive methods are the preferred first-line method of analgesia, such as heat/cold therapy, physical therapy, massage, rest, or exercises, etc. Despite evidence supporting their use, these therapies are only sometimes covered by insurance, and access and cost can be barriers, particularly for uninsured persons who have limited resources, no reliable transportation, or live in rural areas where treatments are not available.
When this is insufficient, non-opioid medications, such as Gabapentin, acetaminophen, or nonsteroidal anti-inflammatory drugs (NSAID), should be considered next. Selective antidepressants and anticonvulsant medications may also be effective. Some examples of when these drugs may be appropriate include neuropathic pain, lower back pain, musculoskeletal injuries (including minor pain related to fractures, sprains, strains, tendonitis, and bursitis), dental pain, postoperative pain, and kidney stone pain.
Providers, however, must also consider the risks and benefits of long-term NSAID use because it may also negatively affect a patient’s gastrointestinal and cardiovascular system. Depending on the diagnosis, a patient may also require an invasive or surgical intervention to treat the underlying cause to alleviate pain.
If the patient has pain that does not sufficiently improve with these initial therapy regimens, at that point, opioids will be the next option to be considered.
It does not mean that patients should be required to sequentially “fail” nonpharmacologic and non-opioid pharmacologic therapy or use any specific treatment before proceeding to opioid therapy. Example: A patient for whom NSAIDs are contraindicated has recently sustained a rotator cuff injury and is experiencing moderate pain to the point at which it is disturbing their sleep, and it will be several weeks before they can have surgery.
Recommendation #2: Discuss with the patient realistic treatment goals for pain and overall function.
Ideally, goals include improving quality of life and function, including social, emotional, and physical dimensions. The provider should help guide these patients to realistic expectations based on their diagnosis. This may mean that the patient may anticipate reduced pain levels but not complete elimination of pain. The provider should discuss the expected or typical timeframe where they may need medications.
If medications are anticipated for acute or subacute pain, a discussion about the expected timeframe for pain should be highlighted in the debate. The patient may then better understand if their recovery is progressing. For chronic conditions, the conversation will focus on or may emphasize the overall risks of beginning long-term medication therapy. They may also advise patients, particularly those with irreversible impairment injuries, that they may experience reduced pain but will not regain function. A withdrawal plan will be discussed if opioid therapy is unsuccessful or the risk vs. benefit ratio is no longer balanced.
The second section of recommendations covers the selection of opioids and dosages.
Recommendation #3: Prescribe immediate-release (I.R.) opioids instead of extended-release and long-acting (ER/LA) opioids when starting opioid therapy.
Immediate-release opioids have faster-acting medication with a shorter duration of pain-relieving action. ER/LA opioids should only be used in patients who have received specific dosages of immediate-release opioids daily for at least one week. Providers should reserve ER/LA opioids for severe, continuous pain, for example, individuals with cancer. ER/LA opioids should not be for PRN use. The reason for this recommendation is to reduce the risk of overdose. A patient who does not feel adequate relief or relief fast enough from the ER/LA dose may be more inclined to take additional amounts sooner than recommended, leading to a potential overdose.
Recommendation #4: Prescribe the lowest effective dose.
Dosing strategies include prescribing low doses and increasing doses in small increments. Prescribe the lowest dose for opioid patients. Carefully consider risk vs. benefits when increasing amounts for individuals with subacute and chronic pain who have developed tolerance. Providers should continue to optimize non-opioid therapies while continuing opioid therapy. It may include recommendations for taking non-opioid medications in addition to opioids and non-pharmacologic methods.
Providers should use caution and increase the dosage by the smallest practical amount, especially before increasing the total opioid dosage to 50 or greater morphine milligram equivalent (MME) daily. Increases beyond 50 MME/day are less likely to provide additional pain relief benefits. The greater the dosage increases, the tendency for risk also increases. Some states require providers to implement clinical protocols at specific dosage levels.
Recommendation #5: Tapering opioids includes weighing the benefits and risks when changing the opioid dosage.
Providers should consider tapering to a reduced dosage or tapering and discontinuing therapy and discuss these approaches before initiating changes when:
- The patient requests dosage reduction or discontinuation,
- Pain improves and might indicate the resolution of an underlying cause,
- Therapy has not reduced pain or improved function,
- Treated with opioids for a long time (e.g., years), and the benefit-risk balance is not clear.
- Receiving higher opioid dosages without evidence of improvement.
- Side effects that diminish the quality of life or cause impairment.
- Opioid misuse
- The patient experiences an overdose or severe event.
- Receiving medications or having a condition that may increase the risk of an adverse event.
Opioid therapy should not be discontinued abruptly unless there is a threat of a severe event, and providers should not rapidly reduce opioid dosages from higher dosages.
Patient agreement and interest in tapering will be key components of successful tapers. Integrating behavioral and non-opioid treatment and interventions for comorbid mental health conditions before/during a taper can help manage pain, strengthen the therapeutic relationship between the provider and patient, and improve the likelihood of positive tapering outcomes. When dosages are reduced or discontinued, a taper slow enough to reduce symptoms and withdrawal should be used. Patients should receive education on possible withdrawal symptoms and when to contact the provider.
For those taking opioids for a shorter duration, a 10% decrease of the original dose per week or slower until close to 30% of the initial amount is reached, followed by a weekly reduction of roughly 10% of the remaining dose) is less likely to trigger withdrawal. Tapers of 10% per month or less are better tolerated than rapidly tapering off when patients have been taking opioids for a longer duration (e.g., for a year or longer). Significant opioid withdrawal symptoms can indicate the need to slow the taper rate further. Short-term medications might also help manage withdrawal symptoms. Providers should follow up frequently (at least monthly) with patients engaging in opioid tapering.
Close monitoring is required for patients who cannot taper and who continue on high doses or otherwise high-risk opioid regimens and should collaborate with patients to mitigate overdose risk. Some patients with unanticipated challenges to tapering may need to be evaluated for OUD.
The third section focuses on the duration of opioid therapy and routine patient follow-up.
Recommendation #6: Prescribing no greater quantity than needed for the expected duration of severe pain requiring opioids.
A few days or less is often enough when opioids are used for common causes of nonsurgical acute pain. Many states have passed legislation that limits initial opioid prescriptions for acute pain to less than seven days. Many insurers and pharmacies have enacted similar policies. Providers should avoid prescribing additional opioids to patients if pain continues longer than expected.
Providers should prescribe and advise opioid use only as needed rather than on a scheduled basis (e.g., one tablet every 4 hours). Limiting the duration of therapy can decrease the need to taper. However, tapering may need to be considered if patients take these medications around the clock for more than a few days.
Longer durations of therapy may be needed when the injury is expected to result in prolonged severe pain (e.g., greater than seven days for severe traumatic injuries). Patients should be evaluated at least every two weeks if they are receiving opioids for acute pain. Suppose opioids are continued for a month or longer. In that case, providers should address potentially reversible causes of chronic pain so that the length of therapy does not continue to extend.
Recommendation #7: Evaluate the benefits and harms of opioid therapy regularly.
The benefits and risks for Evaluating benefits and risks within 1-4 weeks of starting long-term opioid therapy for subacute and chronic pain should be evaluated within 1-4 weeks of initiating therapy and after dosage increases.
The evaluation should include patient perspectives on progress and challenges in moving toward treatment goals, including sustained improvement in pain and function. The Three-item Pain, Enjoyment of Life, and General Activity (PEG) assessment scale could be utilized to help determine patient progress.
Providers should also ask patients about common adverse effects, such as constipation and drowsiness, and assess for outcomes that might be early warning signs for more serious problems such as overdose or OUD.
Patient re-evaluation should occur after therapy begins (about two weeks) when ER/LA opioids are prescribed if the total daily opioid dosage is greater than or equal to 50 MME/day or if there is a concurrent benzodiazepine prescription. These individuals are at a higher risk for overdose. Follow-up for individuals starting or increasing the dosage of methadone is recommended every 2-3 days for the first week. Providers should reassess all patients receiving long-term opioid therapy at least every three months.
The last section of recommendations covers patients at risk for OUD and overdose.
Recommendation #8: Use strategies to mitigate risk by evaluating risk for opioid-related harms, discussing risk with patients, and incorporating risk reduction strategies into the treatment plan.
The patient’s habits (including alcohol and illicit drug use) and behavioral and mental health must be considered. Patients with a history of substance use disorders, depression, and/or mental health disorders have a higher risk of overdose and OUD. Even though the dangers of opioid therapy are higher with these patients, they may still require opioid treatment for pain management.
Psychological distress can interfere with the improvement of pain and/or function in patients experiencing chronic pain; using tools like the Generalized Anxiety Disorder (GAD)-7 and the Patient Health Questionnaire (PHQ-9 or PHQ-4) to assess for anxiety, post-traumatic stress disorder (PTSD), and depression might help providers improve overall pain treatment outcomes. They should also ensure that treatment for depression and other mental health conditions is effective, consulting with behavioral health specialists when needed.
Additionally, providers should:
- Educate on the risks of overdose when opioids are combined with other drugs or alcohol.
- Use caution when prescribing opioids for people with sleep-disordered breathing due to their increased risk for respiratory depression. The provider may ascertain if a patient is compliant with prescribed CPAP.
- Use caution and increased monitoring for patients with renal or hepatic insufficiency.
- Use caution and increased monitoring for patients aged 65 years or older.
- Offering naloxone when prescribing opioids, particularly to patients at increased risk for overdose.
If patients experience a nonfatal opioid overdose, providers should evaluate for OUD. Providers should reduce opioid dosage, discontinue opioids when indicated, continue monitoring, and support for patients prescribed or not prescribed opioids.
Recommendation #9: Reviewing prescription drug monitoring program (PDMP) data.
Providers should review PDMP data specifically for prescription opioids, benzodiazepines, and other controlled medications patients have received from additional prescribers to determine all the opioids the patient could potentially receive. Patients with multiple prescriptions and from various providers are at an increased risk for overdose or OUD. PDMP data should be reviewed before initial drugs for subacute or chronic pain and at least every three months during long-term opioid therapy.
Recommendation # 10: Considering the benefits and risks of [urine] toxicology testing.
Toxicology testing should be used to inform and improve patient care. Providers, practices, and health systems should minimize bias in testing and not test based on assumptions about different patients.
Recommendation # 11: Use caution when prescribing opioid pain medication and other medications concurrently.
Benzodiazepines and opioids can cause CNS depression and potentiate opioid-induced decreases in respiratory drive. Because other CNS depressants can potentiate respiratory depression associated with opioids, benefits vs. risks should be considered.
Recommendation # 12: Offering or arranging treatment for OUD if needed.
Includes referring a patient to a specific treatment center where behavior therapy and medications may be prescribed.

Self Quiz
Ask yourself...
- Do you plan to update your prescribing practices with these new guidelines?
- How often do you assess your patients who require subacute or chronic opioid treatment for their response to treatment?
- How often do you suggest tapering or adjusting opioid dosages?
- Do you routinely screen patients for OUD who are receiving chronic therapy?
- What do you tell patients who would significantly benefit from opioid therapy (e.g., post-operative patients) who are afraid to take them due to adverse side effects?
Prescription Drug Monitoring Programs (PDMP) and Electronic Prescribing.
Prescription Drug Monitoring Programs (PDMP) is a database that keeps track of controlled substance prescriptions. It helps to improve opioid prescribing, inform clinical practice, and protect at-risk patients. A pharmacist must enter controlled substances into the state PDMP when dispensing them. When the pharmacist enters this data, it may occur at various intervals, from one month, daily, or even "real-time." However, a PDMP is only helpful if providers check the system before prescribing (10).
Some states have implemented legislation that requires providers to check a state PDMP before prescribing certain controlled substances and in certain circumstances. Most current mandates require that all prescribers query PDMPs when prescribing any opioid. Some states require prescribers to query PDMPs every time a controlled substance is prescribed, while others require a query only for the initial prescription. Subsequent checks of PDMPs also vary from every time a drug is issued to specific intervals (e.g., every 90 days, twice a year, annually) should prescribing continue.
Some mandates have categorical requirements; e.g., a query must be made if the prescription is over a three-day or seven-day supply or if a certain prescribed level of MME is exceeded. Other states' mandates are based on subjective criteria, e.g., a prescriber's judgment of possible inappropriate use or the prescriber's discretion regarding whether to query the PDMP. Finally, some states mandate that only prescribers in opioid treatment programs, workers' compensation programs, or pain clinics must query PDMPs (10, 11).
In addition to PDMPs, there has been an increase in requirements for providers to utilize Electronic Prescribing for Controlled Substances (ECPS). Electronic prescribing programs for both providers and pharmacies must meet DEA requirements. The DEA's March 31, 2010, conditions were updated on July 27, 2023 (12).
On January 1, 2023, the Centers for Medicare and Medicaid Services (CMS) implemented additional requirements for controlled substances for recipients of Medicare Part D. There are over 51 million U.S. people enrolled in Medicare Part D (Center for Medicare Advocacy, 2023). In addition to state laws, these rules require that prescribers e-prescribe at least 70 percent of controlled substances for patients that have Medicare Part D. A waiver may be approved if the prescriber cannot conduct electronic prescribing due to circumstances beyond the provider's control (12).
Starting June 27, 2023, the 'Consolidated Appropriations Act of 2023' requires new or renewing Drug Enforcement Administration (DEA) registrants, to have at least one of the following:
- A total of eight hours of training from specific organizations on opioid or other substance use disorders
- Board certification in addiction medicine or addiction psychiatry from the American Board of Medical Specialties, American Board of Addiction Medicine, or the American Osteopathic Association
- Graduation within five years and status in good standing from medical, advanced practice nursing, or physician assistant school in the U.S. that included an opioid or other substance use disorder curriculum of at least eight hours (11, 12).
Providers must follow either state law or DEA/CMS regulations, whichever is more stringent. The following map indicates various rules in each state, which will continue to change when new legislation is enacted (11, 12).


Self Quiz
Ask yourself...
- What are the laws in your state about PDMP and E-Prescribing?
- What references can you refer to find out prescribing laws in your state?
- Is the use of PDMP a common practice in your workplace?
Important teaching points about opioid storage and disposal.
Safe Storage
Patients should understand that prescription opioids need to be stored securely (i.e., keeping them in a locked area). This is especially true if children, teens, and other visitors in the house may be aware of their presence. Teens and young adults are the biggest misusers of prescription pain medication. In 2018, over 695,000 youths ages 12–17 and 1.9 million young adults ages 18–25 reported misusing prescription pain medication in the past year. Young people may misuse prescription opioids for many reasons, including curiosity, peer pressure, and wanting to fit in. Another reason teens and young adults may decide to take prescription opioids is because they can be easier to get than other drugs. Studies show that 53% percent of people over 12 who obtained prescription pain medication for non-medical use received them from a friend or relative (13).
Safe Disposal
Patients should be advised on how to get rid of unused or expired medications. The best option is to immediately take them to a drug take-back site, location, or program. These sites or programs can be found online, or the pharmacist may have information. If it is not feasible for the patient to get rid of the drug using a take-back program, the patient should be advised to check if it is on the FDA flush list. If it is, the medication should be flushed down the toilet. Again, the list of drugs is available on the FDA website. If it is not on that list, it should be discarded in the trash at home.
Patients should follow these disposal instructions: Mix medicines (liquid or pills; do not crush tablets or capsules) with an unappealing substance such as dirt, cat litter, or used coffee grounds. Next, place the mixture in a container such as a sealed plastic bag; then throw away the container in your trash at home. Last, the patient should remove or permanently cover all personal information on the prescription label of empty medicine bottles or packaging, then trash or recycle the open container (14).

Self Quiz
Ask yourself...
- Do you often teach your patients about the safe storage and disposal of opioids? If not, what are some barriers to providing this education? How could you overcome these barriers?
- Your patient asks if they can give the leftover pills to their spouse, who has back pain; what would be an appropriate response?
- The patient replies that it is the same medication their spouse has been prescribed and does not understand why they cannot share it; what education will you provide?
OUD treatment, including medications.
Treatment for OUD is multi-faceted and typically includes both mental health components and FDA-approved OUD treatment medications. Mental health components may consist of counseling or a structured treatment program. Cognitive behavioral therapy (CBT) may also be beneficial. A potential barrier to OUD treatment, on the provider's and patient's behalf, is the perception that patients must engage in counseling to start or continue receiving OUD treatment medication. While the mental health components are essential, there may be barriers for patients to begin mental health treatment programs, which include expense, travel, and available openings within the programs. Medication therapy may be a helpful start for these patients (9).
FDA-approved medications indicated for the treatment of OUD include methadone, buprenorphine, and naltrexone. Suboxone is a combination drug composed of buprenorphine and naltrexone.
Medication has several advantages as part of the OUD treatment plan.
- Help the individual to remain safe and comfortable during detox.
- Reduce or eliminate cravings for opioids
- Minimize relapse since the individual is not experiencing uncomfortable withdrawal symptoms
- Allow the individual to focus on therapy without being distracted by withdrawal symptoms and cravings
- Increase safety in cases of overdose
Methadone
Methadone is a full agonist opioid and is a Schedule II controlled medication. Methadone can be prescribed purely for the treatment of pain, as well as for OUD. Methadone treatment for OUD can only be provided through a Substance Abuse and Mental Health Services (SAMHSA)-certified opioid treatment program. Patients taking methadone to treat OUD must receive the medication under the supervision of a clinician. After consistent compliance, patients may take methadone at home between program visits. The length of methadone treatment should be a minimum of 12 months. Methadone doses are often adjusted and readjusted. Methadone is slowly excreted, and there is overdose potential if not taken as prescribed (9).
Buprenorphine
Buprenorphine is a partial agonist opioid. Buprenorphine can be prescribed by any provider with a current, standard DEA registration as a Schedule III Controlled Substance. Like opioids, it produces effects such as euphoria or respiratory depression. With buprenorphine, however, these effects are weaker than those of full opioids such as heroin and methadone. It also has unique pharmacological properties that help lower the potential for misuse and diminish the effects of physical dependency opioids, such as withdrawal symptoms and cravings (9). Subutex was a brand-name version of buprenorphine, discontinued in 2011 after new formulations that were less likely to be misused were developed.
Naltrexone
Naltrexone is an opioid antagonist, not addictive, and does not cause withdrawal symptoms. It blocks the euphoric and sedative effects of opioids by binding and blocking opioid receptors and reduces and suppresses opioid cravings. There is no potential for misuse and diversion. Naltrexone can be prescribed in any setting and can be taken as a pill or once monthly extended-release intramuscular injection (9).
It was estimated 2021 that of the 2.5 million people with OUD, only 36% received any treatment, and only 22% received medications. A part of the July 27, 2023, Consolidated Appropriations Act amended the Controlled Substances Act to eliminate the requirement that providers obtain a specific waiver (a DATA waiver) to prescribe buprenorphine (including Suboxone) to treat opioid use disorder, known as the X-waiver. Additionally, there are no longer any caps on the number of patients a practitioner can treat. This, however, does not change the requirements for methadone treatment (15).


Self Quiz
Ask yourself...
- What are some problems that can occur if opioid medications are not managing pain adequately?
- What are some possible ways you can obtain a detailed, patient-centric health history?
- What are some possible ways APRNs can educate patients on pain and opioid medication options?
Case Study #1
Patient Name: John Henderson,
Gender: Male
Age: 60
Height: 6' 1"
Weight: 190 lbs.
He is employed as a grocery store manager. Reports not using tobacco; drinks alcohol occasionally and has no illicit drug use. He has hypertension and high cholesterol and takes Losartan 50mg daily and Atorvastatin 20 mg daily.
This patient presents to a primary care office with pain and stiffness in the shoulder joint, which has progressively worsened for six months following rotator cuff surgery. He states his pain is unchanged, and he has a limited range of motion. It has been interfering with his ability to do his job. He said he went to physical therapy for a few weeks after his surgery but admitted he did not often complete the home exercise program regimen. An MRI was obtained and showed he had adhesive capsulitis. What are treatment options to consider for "frozen shoulder"?
Given the patient's diagnosis, non-opioid therapy options include nonsteroidal anti-inflammatory drugs, intraarticular glucocorticoid injections, steroid injections into the shoulder joint, range of motion exercises, physical therapy, and consulting with an orthopedic specialist who may recommend a joint manipulation under anesthesia. Each of these should be considered before opioid therapy.
Nursing Considerations
Nurses remain the most trusted profession for a reason, and advanced practice registered nurses (APRNs) are often pillars of patient care in several health care settings. Patients turn to nurses for guidance, education, and support. While there are no specific guidelines for the nurse’s role in opioid education and management, here are some suggestions to provide quality care for patients currently taking opioid medications.
Obtain a Detailed Health History
Often times, pain and mental health can be dismissed and overlooked in health care settings. If a patient is complaining of symptoms that could be related to pain, inquire more about that complaint. Ask about how long the symptoms have lasted, what treatments have been tried, if these symptoms interfere with their quality of life, and if anything alleviates any of these symptoms. If you feel like a patient's complaint is not being taken seriously by other health care professionals, advocate for that patient to the best of your abilities. A detailed pain assessment and history can provide context for opioid pain management and a patient's plan of care. When taking a health history, ask about any prior surgeries, major life stressors in the past year, or any prior opioid usage.
Review the Medication History
Often times, in busy clinical settings, reviewing health records can be overwhelming. Many people take pain medications, including opioids, for various reasons. Ask patients how they are feeling on the medication, if their symptoms are improving, if there are any changes to medication history, and if they use any other substances other than prescribed medications, such as alcohol, tobacco, or other drugs. Remember, prescription medications are not the only medications people take. Confirm medication route, dosage, frequency, and all the details to make sure you and the patient are on the same page and to avoid medication errors and complications. Medication history should be reviewed at every encounter.
Avoid Making Judgements
Society often stigmatizes open discussions of prescription medication and pain. Patients may avoid asking for pain medication for fear of being perceived as a "drug seeker." Other times, patients may have OUD and continuously ask for an increased number of opioid medications. Be willing to be honest with yourself about your comfort level discussing topics and providing education on opioid medications, drug interactions, and pain management. Be willing to address any questions/concerns the patients may have without making judgements.
Communicate the Plan of Care
Communicate the plan of care to other staff involved for continuity of care. For several patients, especially for patients with chronic pain or who use opioids long-term, care often involves a team of mental health professionals, physical therapists, nurses, specialists, pharmacies, and more. Ensure that patients' records are up to date for ease in record sharing and continuity of care and to reduce the incidence of opioid medication errors.
Engage in Self Learning
Stay up to date on continuing education related to opioid medications, pain management, and prescribing regulations. Evidence-based information and scope of practice is always evolving and changing. You can then present your new learning and findings to other health care professionals and educate your patients with the latest information. You can learn more about the latest research on pain management medications, non-pharmacological pain management options, and opioids by following updates from evidence-based organizations, such as the CDC or your local health department.
Perform Pain Assessments
As we know, it is not possible to look at someone with the naked eye and determine if they are in pain. Sometimes, it may be obvious when a patient is in pain (e.g., visible lacerations) and need pain management options, such as opioids. Other times, pain management is addressed as a result of taking a complete health history, listening to patient's concerns, completing a pain assessment, and offering testing to determine the cause of pain.
Assess for Opioid Use Disorder
While it is not possible to look at someone and determine if they have OUD, APRNs should pay attention to certain behaviors, for example, when a patient continually asks for more opioid medications or mentions that they are experiencing many symptoms common to those of OUD. OUD may be diagnosed as a result of completing a health history, listening to patient's concerns, and offering testing to determine the cause of pain. Remember, anyone can have OUD, and no two OUD patients appear the same.
Provide Patient Teaching
Patients should know that anyone has the possibility of experiencing side effects of opioid medications, just like with any medication. Patients should be aware that if they notice any changes in their breathing, changes in their heart rate, or feel like something is a concern, they should seek medical care. Because of social stigma associated with opioids and pain management, people may be hesitant to seek medical care because of fear, shame, and embarrassment. However, as more research and social movements discuss opioid use, there is more space and awareness for opioid education and opioid overdose prevention.
Nurses should also teach patients to advocate for their own health in order to avoid possible opioid complications and poor pain management.
Here are important tips for patient education in the inpatient or outpatient setting.
- Tell the health care provider of any existing medical conditions or concerns (need to identify risk factors)
- Tell the health care provider of any existing lifestyle concerns, such as alcohol use, other drug use, sleeping habits, diet, menstrual cycle changes (need to identify lifestyle factors that can influence opioid use and pain management)
- Tell the health care provider of any prior experiences with opioid medication (if applicable) and any medication reactions or side effects (need to identify risk factors, address pain management appropriately, identify any allergies, and avoid possible opioid overdose symptoms)
- Tell the health care provider if you have any changes in your breathing, bodily functions, or heart rate (potential opioid overdose symptoms)
- Tell the nurse or health care provider if you experience any pain that increasingly becomes more severe or interferes with your quality of life
- Keep track of your pain, overall health, medication use, and health concerns via an app, diary, or journal (self-monitoring for any changes)
- Tell the health care provider right away if you are having thoughts of hurting yourself or others (possible increased risk of suicidality and public safety concerns)
- Take all prescribed medications as indicated and ask questions about medications and possible other treatment options, such as non-pharmacological options or surgeries
- Tell the health care provider if you notice any changes while taking medications or other treatments to manage your pain (potential worsening or improving health situation)
Research
There is extensive publicly available literature on opioids medications. These can be found via the National Institutes of Health website and other evidence-based journals. As research is dependent upon the available of study participants, there are several ways people who take opioids can become part of research. If a patient is interested in participating in clinical trial research, APRNs can encourage them to seek more information on clinical trials from local universities and health care organizations.
Case Study #2
- Patient: Pilar
- Age: 40
- Height: 5' 1"
- Weight: 135 lbs.
Pilar presents to the urgent care clinic today complaining of a severe migraine, which started yesterday. Her history includes a hip injury following a car accident three years ago in which she developed chronic post-traumatic arthritis in the hip. After a total hip arthroplasty, she was diagnosed with heterotopic ossification (bone grows in tissue where it shouldn’t). For the past year, she has been taking 20 mg of oxycodone twice daily to manage chronic hip pain after unsuccessful non-opioid therapies. She has three children with no known pregnancy or postpartum complications. She previously worked part-time as an administrative assistant but has been off work since the car accident. She has post-traumatic stress disorder (PTSD) related to the accident. She has been prescribed Xanax 0.5mg up to three times daily for anxiety. She does not smoke, drink alcohol, or take illicit substances.
- What are some specific questions you'd want to ask about the hip arthritis?
- What are some health history questions you'd want to highlight?
- What lab work or testing would you suggest to perform?
For her migraine, Pilar stated she needed to take more of her oxycodone to deal with the pain and because she could not sleep last night. She is concerned because she is running out of pills. She said her primary care doctor's office was closed, so she came to the urgent care.
- What are some non-pharmacological interventions you can do for Pilar's pain?
- What are some questions you'd want to ask about her migraine?
- What are side effects of opioids would you discuss with her?
Here are some things to consider for Case Study #2.
- Discuss concerns with the patient. This includes taking the opioid more often than prescribed, potential problems with respiratory depression, and overdose.
- Recommend trying eletriptan or dihydroergotamine nasal spray first rather than additional opioids.
- Review the PDMP to see the prescription history for this patient. Attempt to contact the primary care provider to develop a plan of care.
- Consider conducting toxicology testing
- Consider offering naloxone
- Use the DSM-5 criteria to assess the presence (and severity) of OUD or arrange an assessment with a substance use disorder specialist. Offer treatment for OUD if it is confirmed.
Case Study #3
Sabrina is a 16-year-old Black high school student working as a waitress at a local restaurant. She arrives to the local pediatric emergency room after her shift with her mom because she thinks she is experiencing a sickle cell crisis. Sabrina reports that she has these crises every few months, and this is probably the third time she's been in this much pain. She reports being at this same ER last year for something similar. Her mother is completing paperwork and would like Sabrina to get some pain medication as well.
- What are some specific questions you'd want to ask about her health?
- What are some health history questions you'd want to highlight?
- What lab work or testing would you suggest to perform?
- What pain assessments would you perform on Sabrina?
Sabrina agrees to provide bloodwork, complete imaging, and be admitted. She said that no health care provider talked to her about how painful sickle cell crises can be, and she doesn't routinely take pain medication because she "doesn't want to be addicted." Sabrina and her mom heard about pain management options for these extremely painful episodes from social media and the internet and would like Sabrina to get her pain controlled. Sabrina said that she had some opioids last time she was in the ER, but she doesn't remember the name. Her mom doesn't remember the name either, but she remembers it was in an IV medication.
- How would you discuss Sabrina's pain management concerns?
- Given Sabrina's age, medical history, and prior history of opioid use, what medication options would be appropriate for a sickle cell crisis in an adolescent?
Sabrina has been in the pediatric ER for over a day receiving IV hydromorphone. She reports some relief, but Sabrina and her mom are concerned. Sabrina wants to live her life like a normal teenager without being in the hospital every few months for pain. Her mom asks if there is a way to have pain medication at home. Both Sabrina and her mom would like to know if there is anything that can be done to help with the pain outside of medications as well. Sabrina doesn't want to use pain medications daily but wants to have them at home just in case she can't get to the hospital.
- Knowing Sabrina's concerns, what are some possible non-pharmacological pain management options?
- Knowing Sabrina's health history, what would be some patient education talking points about at-home opioid medications and possible side effects?
- What are some possible consequences of leaving pain improperly managed?

Final Reflection Questions
- Are you familiar with any current research on opioid use?
- What are some reasons someone would want to enroll in clinical trials?
- How can nurses make a contribution to research?
- Do you plan to update your prescribing practices to reflect the new CDC guidelines?
Conclusion
Even providers who do not prescribe opioids should be familiar with the effects of opioids and OUD due to its high prevalence in the United States. Understanding the types of pain, how pain occurs, and how it impacts a person's quality of life is especially important. There is still an associated stigma among patients who use Opioids to treat chronic pain conditions. It is essential to recognize that there are times when opioid use is appropriate, as long as the provider practices the recommended guidelines and sound clinical judgment.
Controlled Substances
Introduction
Pain is complex and subjective. The experience of pain can significantly impact an individual's quality of life. According to the National Institute of Health (NIH) (40), pain is the most common complaint in a primary care office, with 20% of all patients reporting pain. Chronic pain is the leading cause of disability, and effective pain management is crucial to health and well-being, particularly when it improves functional ability. Effective pain treatment starts with a comprehensive, empathic assessment and a desire to listen and understand. Nurse Practitioners are well-positioned to fill a vital role in providing comprehensive and empathic patient care, including pain management (23).
While the incidence of chronic pain has remained a significant problem, how clinicians manage pain has significantly changed in the last decade, primarily due to the opioid epidemic. This education aims to discuss pain and the assessment of pain, federal guidelines for prescribing, the opioid epidemic, addiction and diversion, and recommendations for managing pain.
Definition of Pain
Understanding the definition of pain, differentiating between various types of pain, and recognizing the descriptors patients use to communicate their pain experiences are essential for Nurse practitioners involved in pain management. By understanding the medical definition of pain and how individuals may communicate it, nurse practitioners can differentiate varying types of pain to target assessment.
According to the International Association for the Study of Pain (27), pain is "an unpleasant sensory and emotional experience associated with actual or potential tissue damage or terms of described such in damage." The IASP, in July 2020, expanded its definition of pain to include context further.
Their expansion is summarized below:
- Pain is a personal experience influenced by biological, psychological, and social factors.
- Pain cannot be inferred solely from activity in sensory neurons.
- Individuals learn the concept of pain through their life experiences.
- A person's report of an experience in pain should be respected.
- Pain usually serves an adaptive role but may adversely affect function and social and psychological well-being.
- The inability to communicate does not negate the possibility of the experience of pain.


Self Quiz
Ask yourself...
- Analyze how changes to the definition of pain may affect your practice.
- Discuss how you manage appointment times, knowing that 20% of your scheduled patients may seek pain treatment.
- How does the approach to pain management change in the presence of a person with a disability?
Types of Pain
Pain originates from different mechanisms, causes, and areas of the body. As a nurse practitioner, understanding the type of pain a patient is experiencing is essential for several reasons (23).
- Determining an accurate diagnosis. This kind of pain can provide valuable clues to the underlying cause or condition.
- Creating a treatment plan. Different types of pain respond better to specific treatments or interventions.
- Developing patient education. A nurse practitioner can provide targeted education to patients about their condition, why they may experience the pain as they do, its causes, and treatment options. Improving the patient's knowledge and control over their condition improves outcomes.
Acute Pain
Acute pain is typically short-lived and is a protective response to an injury or illness. Patients are usually able to identify the cause. This type of pain resolves as the underlying condition improves or heals (12).
Chronic Pain
Chronic pain is diagnosed when it continues beyond the expected healing time. Pain is defined as chronic when it persists for longer than three months. It may result from an underlying disease or injury or develop without a clear cause. Chronic pain often significantly impacts a person's physical and emotional well-being, requiring long-term management strategies. The prolonged experience of chronic pain usually indicates a central nervous system component of pain that may require additional treatment. Patients with centralized pain often experience allodynia or hyperalgesia (12).
Allodynia is pain evoked by a stimulus that usually does not cause pain, such as a light touch. Hyperalgesia is the effect of a heightened pain response to a stimulus that usually evokes pain (12).
Nociceptive Pain
Nociceptive pain arises from activating peripheral nociceptors, specialized nerve endings that respond to noxious stimuli. This type of pain is typically associated with tissue damage or inflammation and is further classified into somatic and visceral pain subtypes.
Somatic pain is most common and occurs in muscles, skin, or bones; patients may describe it as sharp, aching, stiffness, or throbbing.
Visceral pain occurs in the internal organs, such as indigestion or bowel spasms. It is more vague than somatic pain; patients may describe it as deep, gnawing, twisting, or dull (12).
Neuropathic pain
Neuropathic pain is a lesion or disease of the somatosensory nervous system. Examples include trigeminal neuralgia, painful polyneuropathy, postherpetic neuralgia, and central poststroke pain (10).
Neuropathic pain may be ongoing, intermittent, or spontaneous pain. Patients often describe neuropathic pain as burning, prickling, or squeezing quality. Neuropathic pain is a common chronic pain. Patients commonly describe allodynia and hyperalgesia as part of their chronic pain experience (10).
Affective pain
Affective descriptors reflect the emotional aspects of pain and include terms like distressing, unbearable, depressing, or frightening. These descriptors provide insights into the emotional impact of pain on an individual's well-being (12).

Self Quiz
Ask yourself...
- How can nurse practitioners effectively elicit patient descriptors to accurately assess the type of pain the patient is experiencing?
- Expand on how pain descriptors can guide interventions even if the cause is not yet determined.
- What strategies ensure patients feel comfortable describing their pain, particularly regarding subjective elements such as quality and location?
Case Study
Mary Adams is a licensed practical nurse who has just relocated to town. Mary will be the utilization review nurse at a local long-term care facility. Mary was diagnosed with Postherpetic Neuralgia last year, and she is happy that her new job will have her mostly doing desk work and not providing direct patient care as she had been before the relocation. Mary was having difficulty at work at her previous employer due to pain. She called into work several times, and before leaving, Mary's supervisor had counseled her because of her absences.
Mary wants to establish primary care immediately because she needs ongoing pain treatment. She is hopeful that, with her new job and pain under control, she will be able to continue a successful career in nursing. When Mary called the primary care office, she specifically requested a nurse practitioner as her primary care provider because she believes that nurse practitioners tend to spend more time with their patients.
Assessment
The assessment effectively determines the type of treatment needed, the options for treatment, and whether the patient may be at risk for opioid dependence. Since we know that chronic pain can lead to disability and pain has a high potential to negatively affect the patient's ability to work or otherwise, be productive, perform self-care, and potentially impact family or caregivers, it is imperative to approach the assessment with curiosity and empathy. This approach will ensure a thorough review of pain and research on pain management options. Compassion and support alone can improve patient outcomes related to pain management (23).
Record Review
Regardless of familiarity with the patient, reviewing the patient's treatment records is essential, as the ability to recall details is unreliable. Reviewing the records can help identify subtle changes in pain description and site, the patient's story around pain, failed modalities, side effects, and the need for education, all impacting further treatment (23).
Research beforehand the patient's current prescription and whether or not the patient has achieved the maximum dosage of the medication. Analysis of the patient's past prescription could reveal a documented failed therapy even though the patient did not receive the maximum dose (23).
A review of documented allergens may indicate an allergy to pain medication. Discuss with the patient the specific response to the drug to determine if it is a true allergy, such as hives or anaphylaxis, or if the response may have been a side effect, such as nausea and vomiting.
Research whether the patient tried any non-medication modalities for pain, such as physical therapy (PT), occupational therapy (OT), or Cognitive Behavioral Therapy (CBT). Note any non-medication modalities documented as failed therapies. The presence of any failed therapies should prompt further discussion with the patient, family, or caregiver about the experience. The incompletion of therapy should not be considered failed therapy. Explore further if the patient abandoned appointments.
Case Study
You review the schedule for the week, and there are three new patient appointments. One is Mary Adams. The interdisciplinary team requested and received Mary's treatment records from her previous primary care provider. You make 15 minutes available to review Mary's records and the questionnaire Mary filled out for her upcoming appointment. You see that Mary has been diagnosed with Postherpetic Neuralgia and note her current treatment regimen, which she stated was ineffective. You write down questions you will want to ask Mary. You do not see evidence of non-medication modalities or allergies to pain medication.

Self Quiz
Ask yourself...
- What potential risks or complications can arise from neglecting to conduct a thorough chart review before initiating a pain management assessment?
- In your experience, what evidence supports reviewing known patient records?
- What is an alternative to reviewing past treatment if records are not available?
Pain Assessment
To physically assess pain, several acronyms help explore all the aspects of the patient's experience. Acronyms commonly used to assess pain are SOCRATES, OLDCARTS, and COLDERAS. These pain assessment acronyms are also helpful in determining treatment since they include a character and duration of pain assessment (23).
| O-Onset | S-Site | C-Character |
| L-Location | O-Onset | O-Onset |
| D-Duration | C-Character | L-Location |
| C-Character | R-Radiate | D-Duration |
| A-Alleviating | A-Associated symptoms | E-Exacerbating symptoms |
| R-Radiating, relieving | T-Time/Duration | R-Relieving, radiating |
| T-Temporal patterns (frequency) | E-Exacerbating | A-Associated symptoms |
| S-Symptoms | S-Severity | S-Severity of illness |
Inquire where the patient is feeling pain. The patient may have multiple areas and types of pain. Each type and location must be explored and assessed. Unless the pain is from a localized injury, a body diagram map, as seen below, is helpful to document, inform, and communicate locations and types of pain. In cases of Fibromyalgia, rheumatoid arthritis, or other centralized or widespread pain, it is vital to inquire about radiating pain. The patient with chronic pain could be experiencing acute pain or a new pain site, such as osteoarthritis, that may need further evaluation and treatment (23).
Inquire with the patient how long their pain has been present and any associated or known causative factors. Pain experienced longer than three months defines chronic versus acute pain. Chronic pain means that the pain is centralized or a function of the Central Nervous system, which should guide treatment decisions.
To help guide treatment, ask the patient to describe their pain. The description helps identify what type of pain the patient is experiencing: Allodynia and hyperalgesia indicate centralized pain; sharp, shooting pain could indicate neuropathic pain. Have the patient rate their pain. There are various tools, as shown below, for pain rating depending on the patient's ability to communicate. Not using the pain rating number alone is imperative. Ask the patient to compare the severity of pain to a previous experience. For example, a 1/10 may be experienced as a bumped knee or bruise, whereas a 10/10 is experienced on the level of a kidney stone or childbirth (23).
Besides the 0-10 rating scale and depending on the patient's needs, several pain rating scales are appropriate. They are listed below.
The 0-5 and Faces scales may be used for all adult patients and are especially effective for patients experiencing confusion.
The Defense and Veterans Pain Rating Scale (DVPRS) is a five-item tool that assesses the impact of pain on sleep, mood, stress, and activity levels (20).
For patients unable to self-report pain, such as those intubated in the ICU or late-stage neurological diseases, the FLACC scale is practical. The FLACC scale was initially created to assess pain in infants. Note: The patient need not cry to be rated 10/10.
| Behavior | 0 | 1 | 2 |
| Face | No particular expression or smile | Occasional grimace or frown, withdrawn, disinterested | Frequent or constant quivering chin, clenched jaw |
| Legs | Normal position or relaxed | Uneasy, restless, tense | Kicking or legs drawn |
| Activity | Lying quietly, in a normal position, or relaxed | Squirming, shifting back and forth, tense | Arched, rigid, or jerking |
| Cry | No cry wake or asleep | Moans or whimpers: occasional complaints | Crying steadily, screams, sobs, frequent complaints |
| Consolability | Content, relaxed | Distractable, reassured by touching, hugging, or being talked to | Difficult to console or comfort |
(21).
Assess contributors to pain such as insomnia, stress, exercise, diet, and any comorbid conditions. Limited access to care, socioeconomic status, and local culture also contribute to the patient's experience of pain (23). Most patients have limited opportunity to discuss these issues, and though challenging to bring up, it is compassionate and supportive care. A referral to social work or another agency may be helpful if you cannot explore it fully.
Assess for substance abuse disorders, especially among male, younger, less educated, or unemployed adults. Substance abuse disorders increase the likelihood of misuse disorder and include alcohol, tobacco, cannabis, cocaine, and heroin (29).
Inquire as to what changes in function the pain has caused. One question to ask is, "Were it not for pain, what would you be doing?" As seen below, a Pain, Enjoyment, and General Activity (PEG) three-question scale, which focuses on function and quality of life, may help determine the severity of pain and the effect of treatment over time.
| What number best describes your pain on average in the past week? 0-10 |
| What number best describes how, in the past week, pain has interfered with your enjoyment of life? 0-10 |
| What number determines how, in the past week, pain has interfered with your general activity? 0-10 |
(21).
Assess family history, mental health disorders, chronic pain, or substance abuse disorders. Each familial aspect puts patients at higher risk for developing chronic pain (23).
Evaluate for mental health disorders the patient may be experiencing, particularly anxiety and depression. The Patient Health Questionnaire (PHQ4) is a four-question tool for assessing depression and anxiety.
In some cases, functional MRI or imaging studies effectively determine the cause of pain and the treatment. If further assessment is needed to diagnose and treat pain, consult Neurology, Orthopedics, Palliative care, and pain specialists (23).
Case Study
You used OLDCARTS to evaluate Mary's pain and completed a body diagram. Mary is experiencing allodynia in her back and shoulders, described as burning and tingling. It is exacerbated when she lifts, such as moving patients at the long-term care facility and, more recently, boxes from her move to the new house. Mary has also been experiencing anxiety due to fear of losing her job, the move, and her new role. She has moved closer to her family to help care for her children since she often experiences fatigue. Mary has experienced a tumultuous divorce in the last five years and feels she is still undergoing some trauma.
You saw in the chart that Mary had tried Gabapentin 300 mg BID for her pain and inquired what happened. Mary explained that her pain improved from 8/10 to 7/10 and had no side effects. Her previous care provider discontinued the medication and documented it as a failed therapy. You reviewed the minimum and maximum dosages of Gabapentin and know Mary can take up to 1800mg/day.
During the assessment, Mary also described stiffness and aching in her left knee. She gets a sharp pain when she walks more than 500 steps, and her knee is throbbing by the end of the day. Mary rated the pain a 10/10, but when she compared 10/10 to childbirth, Mary said her pain was closer to 6/10. Her moderate knee pain has reduced Mary's ability to exercise. She used to like to take walks. Mary stated she has had knee pain for six months and has been taking Ibuprofen 3 – 4 times daily.
Since Mary's pain is moderate, you evaluate your options of drugs for moderate to severe pain.


Self Quiz
Ask yourself...
- How do you assess and evaluate a patient's pain level?
- What are the different types of pain and their management strategies?
- How do you determine the appropriate dosage of pain medications for a patient?
- How do you assess the effectiveness of pain medications in your patients?
- How do you adjust medication dosages for elderly patients with pain or addiction?
- How do you address the unique challenges in pain management for pediatric patients?
- What is the role of non-pharmacological interventions in pain management?
- How do you incorporate non-pharmacological interventions into your treatment plans?
Opioid Classifications and Drug Schedules
A comprehensive understanding of drug schedules and opioid classifications is essential for nurse practitioners to ensure patient safety, prevent drug misuse, and adhere to legal and regulatory requirements. Nurse practitioners with a comprehensive understanding of drug schedules and opioid classifications can effectively communicate with colleagues, ensuring accurate medication reconciliation and facilitating interdisciplinary care. Nurse practitioners’ knowledge in facilitating discussions with pharmacists regarding opioid dosing, potential interactions, and patient education is essential (49).
Drug scheduling became mandated under the Controlled Substance Act. The Drug Enforcement Agency (DEA) Schedule of Controlled Drugs and the criteria and common drugs are listed below.
|
Schedule |
Criteria | Examples |
|
I |
No medical use; high addiction potential |
Heroin, marijuana, PCP |
|
II |
Medical use; high addiction potential |
Morphine, oxycodone, Methadone, Fentanyl, amphetamines |
|
III |
Medical use; high addiction potential |
Hydrocodone, codeine, anabolic steroids |
|
IV |
Medical use, low abuse potential |
Benzodiazepines, meprobamate, butorphanol, pentazocine, propoxyphene |
| V | Medical use; low abuse potential |
Buprex, Phenergan with codeine |
(Pain Physician, 2008)
Listed below are drugs classified by their schedule and mechanism of action. "Agonist" indicates a drug that binds to the opioid receptor, causing pain relief and also euphoria. An agonist-antagonist indicates the drug binds to some opioid receptors but blocks others. Mixed antagonist-agonist drugs control pain but have a lower potential for abuse and dependence than agonists (7).
| Schedule I | Schedule II | Schedule III | Schedule IV | Schedule V | |
| Opioid agonists |
BenzomorphineDihydromor-phone, Ketobemidine, Levomoramide, Morphine-methylsulfate, Nicocodeine, Nicomorphine, Racemoramide |
Codeine, Fentanyl, Sublimaze, Hydrocodone, Hydromorphone, Dilaudid, Meperidine, Demerol, Methadone, Morphine, Oxycodone, Endocet, Oxycontin, Percocet, Oxymorphone, Numorphan |
Buprenorphine Buprenex, Subutex, Codeine compounds, Tylenol #3, Hydrocodone compounds, Lortab, Lorcet, Tussionex, Vicodin |
Propoxyphene, Darvon, Darvocet | Opium, Donnagel, Kapectolin |
| Mixed Agonist -Antagonist | BuprenorphineNaloxone, Suboxone |
Pentazocine, Naloxone, Talwin-Nx |
|||
| Stimulants | N-methylampheta-mine 3, 4-methylenedioxy amphetamine, MDMA, Ecstacy | Amphetamine, Adderal, Cocaine, Dextroamphetamine, Dexedrine, Methamphetamine, Desoxyn, Methylphenidate, Concerta, Metadate, Ritalin, Phenmetrazine, Fastin, Preludin | Benapheta-mine, Didrex, Pemolin, Cylert, Phendimetra-zine, Plegine | Diethylpropion, Tenuate, Fenfluramine, Phentermine Fastin | 1-dioxy-ephedrine-Vicks Inhaler |
| Hallucinogen-gens, other | Lysergic Acid Diamine LSD, marijuana, Mescaline, Peyote, Phencyclidine PCP, Psilocybin, Tetrahydro-cannabinol | Dronabinol, Marinol | |||
| Sedative Hypnotics |
Methylqualine, Quaalude, Gamma-hydroxy butyrate, GHB
|
Amobarbitol, Amytal, Glutethamide, Doriden, Pentobarbital, Nembutal, Secobarbital, Seconal |
Butibarbital. Butisol, Butilbital, Florecet, Florinal, Methylprylon, Noludar |
Alprazolam, Xanax, Chlordiazepoxide, Librium, Chloral betaine, Chloral hydrate, Noctec, Chlorazepam, Clonazepam, Klonopin, Clorazopate, Tranxene, Diazepam, Valium, Estazolam, Prosom, Ethchlorvynol, Placidyl, Ethinamate, Flurazepam, Dalmane, Halazepam, Paxipam, Lorazepam, Ativan, Mazindol, Sanorex, Mephobarbital, Mebaral, Meprobamate, Equanil, Methohexital, Brevital Sodium, Methyl-phenobarbital, Midazolam, Versed, Oxazepam, Serax, Paraldehyde, Paral, Phenobarbital, Luminal, Prazepam, Centrax, Temazepam, Restoril, Triazolam, Halcion, Sonata, Zolpidem, Ambien |
Diphenoxylate preparations, Lomotil |
(41).

Self Quiz
Ask yourself...
- What are the potential risks and benefits of using opioids for pain management?
- How can nurse practitioners effectively monitor patients on long-term opioid therapy?
- What are the potential risks and benefits of using long-acting opioids for chronic pain?
- How do you monitor patients on long-acting opioids for safety and efficacy?
Commonly Prescribed Opioids, Indications for Use, and Typical Side Effects
Opioid medications are widely used for managing moderate to severe pain. Referencing NIDA (2023), this section aims to give healthcare professionals an overview of the indications and typical side effects of commonly prescribed Schedule II opioid medications, including hydrocodone, oxycodone, morphine, Fentanyl, and hydromorphone.
Opioids are derived and manufactured in several ways. Naturally occurring opioids come directly from the opium poppy plant. Synthetic opioids are manufactured by chemically synthesizing compounds that mimic the effects of a natural opioid. Semi-synthetic is a mix of naturally occurring and man-made (35).
Understanding the variations in how an opioid is derived and manufactured is crucial in deciding the type of opioid prescribed, as potency and analgesic effects differ. Synthetic opioids are often more potent than naturally occurring opioids. Synthetic opioids have a longer half-life and slower elimination, affecting the duration of action and timing for dose adjustments. They are also associated with a higher risk of abuse and addiction (38).
Hydrocodone
Mechanism of Action and Metabolism
Hydrocodone is a Schedule II medication. It is an opioid agonist and works as an analgesic by activating mu and kappa opioid receptors located in the central nervous system and the enteric plexus of the bowel. Agonist stimulation of the opioid receptors inhibits nociceptive neurotransmitters' release and reduces neuronal excitability (17).
- Produces analgesia.
- Suppresses the cough reflex at the medulla.
- Causes respiratory depression at higher doses.
Hydrocodone is indicated for treating severe pain after nonopioid therapy has failed. It is also indicated as an antitussive for nonproductive cough in adults over 18.
Available Forms
Hydrocodone immediate release (IR) reaches maximum serum concentrations in one hour with a half-life of 4 hours. Extended-release (ER) Hydrocodone reaches peak concentration at 14-16 hours and a half-life of 7 to 9 hours. Hydrocodone is metabolized to an inactive metabolite in the liver by cytochrome P450 enzymes CYP2D6 and CYP3A4. Hydrocodone is converted to hydromorphone and is excreted renally. Plasma concentrations of hydromorphone are correlated with analgesic effects rather than hydrocodone.
Hydrocodone is formulated for oral administration into tablets, capsules, and oral solutions. Capsules and tablets should never be crushed, chewed, or dissolved. These actions convert the extended-release dose into immediate release, resulting in uncontrolled and rapid release of opioids and possible overdose.
Dosing and Monitoring
Hydrocodone IR is combined with acetaminophen or ibuprofen. The dosage range is 2.5mg to 10mg every 4 to 6 hours. If formulated with acetaminophen, the dosage is limited to 4gm/day.
Hydrocodone ER is available as tablets and capsules. Depending on the product, the dose of hydrocodone ER formulations in opioid-naïve patients is 10 to 20 mg every 12 to 24 hours.
Nurse practitioners should ensure patients discontinue all other opioids when starting the extended-release formula.
Side Effects and Contraindications
Because mu and kappa opioid receptors are in the central nervous system and enteric plexus of the bowel, the most common side effects of hydrocodone are constipation and nausea (>10%).
Other adverse effects of hydrocodone include:
- Respiratory: severe respiratory depression, shortness of breath
- Cardiovascular: hypotension, bradycardia, peripheral edema
- Neurologic: Headache, chills, anxiety, sedation, insomnia, dizziness, drowsiness, fatigue
- Dermatologic: Pruritus, diaphoresis, rash
- Gastrointestinal: Vomiting, dyspepsia, gastroenteritis, abdominal pain
- Genitourinary: Urinary tract infection, urinary retention
- Otic: Tinnitus, sensorineural hearing loss
- Endocrine: Secondary adrenal insufficiency (17)
Hydrocodone, being an agonist, must not be taken with other central nervous system depressants as sedation and respiratory depression can result. In formulations combined with acetaminophen, hydrocodone can increase the international normalized ratio (INR) and cause bleeding. Medications that induce or inhibit cytochrome enzymes can lead to wide variations in absorption.
The most common drug interactions are listed below:
- Alcohol
- Benzodiazepines
- Barbiturates
- other opioids
- rifampin
- phenytoin
- carbamazepine
- cimetidine,
- fluoxetine
- ritonavir
- erythromycin
- diltiazem
- ketoconazole
- verapamil
- Phenytoin
- John’s Wort
- Glucocorticoids
Considerations
Use with caution in the following:
- Patients with Hepatic Impairment: Initiate 50% of the usual dose
- Patients with Renal Impairment: Initiate 50% of the usual dose
- Pregnancy: While not contraindicated, the FDA issued a black-boxed warning since opioids cross the placenta, and prolonged use during pregnancy may cause neonatal opioid withdrawal syndrome (NOWS).
- Breastfeeding: Infants are susceptible to low dosages of opioids. Non-opioid analgesics are preferred.
Pharmacogenomic: Genetic variants in hydrocodone metabolism include ultra-rapid, extensive, and poor metabolizer phenotypes. After administration of hydrocodone, hydromorphone levels in rapid metabolizers are significantly higher than in poor metabolizers.
Oxycodone
Mechanism of Action and Metabolism
Oxycodone has been in use since 1917 and is derived from Thebaine. It is a semi-synthetic opioid analgesic that works by binding to mu-opioid receptors in the central nervous system. It primarily acts as an agonist, producing analgesic effects by inhibiting the transmission of pain signals (Altman, Clark, Huddart, & Klein, 2018).
Oxycodone is primarily metabolized in the liver by CYP3A4/5. It is metabolized in the liver to noroxycodone and oxymorphone. The metabolite oxymorphone also has an analgesic effect and does not inhibit CYP3A4/5. Because of this metabolite, oxycodone is more potent than morphine, with fewer side effects and less drug interactions. Approximately 72% of oxycodone is excreted in urine (Altman, Clark, Huddart, & Klein, 2018).
Available Forms
Oxycodone can be administered orally, rectally, intravenously, and as an epidural. For this sake, we will focus on immediate-release and extended-release oral formulations.
- Immediate-release (IR) tablets
- IR capsules
- IR oral solutions
- Extended-release (ER) tablets
Dosing and Monitoring
The dosing of oxycodone should be individualized based on the patient's pain severity, previous opioid exposure, and response. Initial dosages for opioid naïve patients range from 5-15 mg for immediate-release formulations, while extended-release formulations are usually initiated at 10-20 mg. Dosage adjustments may be necessary based on the patient's response, but caution should be exercised. IR and ER formulations reach a steady state at 24 hours and titrating before 24 hours may lead to overdose.
Regular monitoring is essential to assess the patient's response to treatment, including pain relief, side effects, and signs of opioid misuse or addiction. Monitoring should include periodic reassessment of pain intensity, functional status, and adverse effects (Altman, Clark, Huddart, & Klein, 2018).
Side Effects and Contraindications
Common side effects of oxycodone include:
- constipation
- nausea
- sedation
- dizziness
- respiratory depression
- respiratory arrest
- hypotension
- fatal overdose
Oxycodone is contraindicated in patients with known hypersensitivity to opioids, severe respiratory depression, paralytic ileus, or acute or severe bronchial asthma. It should be used cautiously in patients with a history of substance abuse, respiratory conditions, liver or kidney impairment, and those taking other medications that may interact with opioids, such as alcohol (4).
It is also contraindicated with the following medications and classes:
- Antifungal agents
- Antibiotics
- Rifampin
- Carbamazepine
- Fluoxetine
- Paroxetine
Considerations
- Nurse practitioners should consider the variations in the mechanism of action for the following:
- Metabolism differs between males and females: females have been shown to have less concentration of oxymorphone and more CYP3A4/5 metabolites.
- Infants have reduced clearance of oxycodone, increasing side effects.
- Pediatrics have 20-40% increased clearance over adults.
- Reduced clearance with age increases the half-life of oxycodone.
- Pregnant women have a greater clearance and reduced half-life.
- Impairment of the liver reduces clearance.
- Cancer patients with cachexia have increased exposure to oxycodone and its metabolite.
- Maternal and neonate concentrations are similar, indicating placenta crossing (4)
Morphine

Mechanism of Action and Metabolism
Morphine is a naturally occurring opioid alkaloid extracted from the opium poppy. It was isolated in 1805 and is the opioid against which all others are compared. Morphine binds to mu-opioid receptors in the brain and spinal cord, inhibiting the transmission of pain signals and producing analgesia. It is a first-line choice of opioid for moderate to severe acute, postoperative, and cancer-related pain (8).
Morphine undergoes first-pass metabolism in the liver and gut. It is well absorbed and distributed throughout the body. Its main metabolites are morphine-3-glucuronide and morphine-6-glucuronide. Its mean plasma elimination half-life after intravenous administration is about 2 hours. Approximately 90% of morphine is excreted in the urine within 24 hours (8).
Available Forms
Morphine is available in various forms, including.
- immediate-release tablets
- extended release tablets
- oral IR solutions
- injectable solutions
- transdermal patches
Dosing and Monitoring
Morphine is hydrophilic and, as such, has a slow onset time. The advantage of this is that it is unlikely to cause acute respiratory depression even when injected. However, because of the slow onset time, there is more likelihood of morphine overdose due to the ability to “stack” doses in patients experiencing severe pain (Bistas, Lopez-Ojeda, & Ramos-Matos, 2023).
The dosing of morphine depends on the patient's pain severity, previous opioid exposure, and other factors. It is usually initiated at a low dose and titrated upwards as needed. Monitoring pain relief, adverse effects, and signs of opioid toxicity is crucial. Reevaluate benefits and harms with patients within 1 to 4 weeks of starting opioid therapy or of dose escalation. General recommendations for initiating morphine (Bistas, Lopez-Ojeda, & Ramos-Matos, 2023).
Prescribe IR opioids instead of ER opioids.
Prescribe the lowest effective dosage, below 50 Morphine Milligram Equivalents (MME) /day.
Side Effects and Contraindications
Because morphine binds to opioid receptors in the brain and spinal cord, is metabolized in the liver and gut, and has a slow onset, the following side effects are common:
- Constipation
- Nausea
- Vomiting
- Sedation
- Dizziness
- Respiratory depression
- Pruritis
- Sweating
- Dysphoria/Euphoria
- Dry mouth
- Anorexia
- Spasms of urinary and biliary tract
Contraindications of morphine are:
- Known hypersensitivity or allergy to morphine.
- Bronchial asthma or upper airway obstruction
- Respiratory depression in the absence of resuscitative equipment
- Paralytic ileus
- Risk of choking in patients with dysphagia, including infants, children, and the elderly (8)
Concurrent use with other sedating medications: Amitriptyline, diazepam, haloperidol, chlorpromazine
Morphine interacts with the following medications:
- Ciprofloxacin
- Metoclopramide
- Ritonavir
Considerations for Nurse Practitioners
Assess for medical conditions that may pose serious and life-threatening risks with opioid use, such as the following:
- Sleep-disordered breathing, such as sleep apnea.
- Pregnancy
- Renal or hepatic insufficiency
- Age >= 65
- Certain mental health conditions
- Substance use disorder
- Previous nonfatal overdose
Fentanyl
Mechanism of Action and Metabolism
Fentanyl is a synthetic opioid more potent than morphine and was approved in 1968. Fentanyl is an agonist that works by binding to the mu-opioid receptors in the central nervous system. This binding inhibits the transmission of pain signals, resulting in analgesia. Fentanyl is often used for severe pain management, particularly in the perioperative and palliative care settings, or for severe pain in patients with Hepatic failure (8).
It is a mu-selective opioid agonist. However, it can activate other opioid receptors in the body, such as the delta and kappa receptors, producing analgesia. It also activates the Dopamine center of the brain, stimulating relaxation and exhilaration, which is responsible for its high potential for addiction (8).
Indications for fentanyl are as follows:
- Preoperative analgesia
- Anesthesia adjunct
- Regional anesthesia adjunct
- General anesthesia
- Postoperative pain control
- Moderate to severe acute pain (off-label)
Available Forms
- Fentanyl is available in various forms, including:
- transdermal patches
- injectable solutions
- lozenges
- nasal sprays
- oral tablets (8)
Dosing and Monitoring
Fentanyl is metabolized via the CYP3A4 enzyme in the liver. It has a half-life of 3 to 7 hours, and 75% of Fentanyl is excreted in the urine and 9% in feces.
The dosing of fentanyl depends on the route of administration and the patient's needs. For example, transdermal patches are typically applied every 72 hours, while injectable solutions are titrated to achieve the desired analgesic effect. Monitoring should include assessing pain levels, respiratory rate, blood pressure, and sedation scores (8).
Fentanyl is most dosed as follows:
- Post-operative pain control
- 50 to 100 mcg IV/IM every 1 to 2 hours as needed; alternately 0.5 to 1.5 mcg/kg/hour IV as needed. Consider lower dosing in patients 65 and older.
PCA (patient-controlled analgesia): 10 to 20 mcg IV every 6 to 20 minutes as needed; start at the lowest effective dose for the shortest effective duration - refer to institutional protocols (8).
Moderate to severe acute pain (off-label) 1 to 2 mcg/kg/dose intranasally each hour as needed; the maximum dose is 100 mcg. Use the lowest effective dose for the shortest effective duration (8).
Side Effects and Contraindications
Common side effects of fentanyl include:
- respiratory depression
- sedation
- constipation
- nausea
- vomiting
- euphoria
- confusion
- respiratory depression/arrest
- visual disturbances
- dyskinesia
- hallucinations
- delirium
- narcotic ileus
- muscle rigidity
- addiction
- loss of consciousness
- hypotension
- coma
- death (8).
The use of fentanyl is contraindicated in patients in the following situations:
- After operative interventions in the biliary tract, these may slow hepatic elimination of the drug.
- With respiratory depression or obstructive airway diseases (i.e., asthma, COPD, obstructive sleep apnea, obesity hyperventilation, also known as Pickwickian syndrome)
- With liver failure
- With known intolerance to fentanyl or other morphine-like drugs, including codeine or any components in the formulation.
- With known hypersensitivity (i.e., anaphylaxis) or any common drug delivery excipients (i.e., sodium chloride, sodium hydroxide) (8).
Considerations for Nurse Practitioners
Nurse practitioners prescribing fentanyl should thoroughly assess the patient's pain, medical history, and potential risk factors for opioid misuse. They should also educate patients about the proper use, storage, and disposal of fentanyl. It should be used cautiously in patients with respiratory disorders, liver or kidney impairment, or a history of substance abuse. Fentanyl is contraindicated in patients with known hypersensitivity to opioids and those without exposure to opioids.
Alcohol and other drugs, legal or illegal, can exacerbate fentanyl's side effects, creating multi-layered clinical scenarios that can be complex to manage. These substances, taken together, generate undesirable conditions that complicate the patient's prognosis (8).
Hydromorphone
Mechanism of Action and Metabolism
Hydromorphone is a semi-synthetic opioid derived from morphine. It binds to the mu-opioid receptors in the central nervous system. It primarily exerts its analgesic effects by inhibiting the release of neurotransmitters involved in pain transmission, thereby reducing pain perception. Hydromorphone also exerts its effects centrally at the medulla level, leading to respiratory depression and cough suppression (1).
Hydromorphone is indicated for:
- moderate to severe acute pain
- severe chronic pain
- refractory cough suppression (off-label) (1)
Available Forms
Hydromorphone is available in various forms, depending on the patient’s needs and severity of pain.
- immediate-release tablet
- extended release tablets
- oral liquid
- injectable solution
- rectal suppositories
Dosing and Monitoring
The immediate-release oral formulations of hydromorphone have an onset of action within 15 to 30 minutes. Peak levels are typically between 30 and 60 minutes with a half-life of 2 to 3 hours. Hydromorphone is primarily excreted through the urine.
The dosing of hydromorphone should be individualized based on the patient's pain intensity, initiated at the lowest effective dose, and adjusted gradually as needed. Close monitoring of pain relief, adverse effects, and signs of opioid toxicity is essential. Patients should be assessed regularly to ensure they receive adequate pain control without experiencing excessive sedation or respiratory depression.
The following are standard dosages that should only be administered when other opioid and non-opioid options fail.
- Immediate-release oral solutions dosage: 1 mg/1 mLoral tablets are available in 2 mg, 4 mg, and 8 mg.
- Extended-release oral tablets are available in dosages of 8 mg, 12 mg, 16 mg, and 32 mg.
- Injection solutions are available in concentrations of 1 mg/mL, 2 mg/mL, 4 mg/mL, and 10 mg/mL.
- Intravenous solutions are available in strengths of 2 mg/1 mL, 2500 mg/250 mL, ten mg/1 mL, and 500 mg/50 mL.
- Suppositories are formulated at a strength of 3 mg (1).
Side Effects and Contraindications
Hydromorphone has potential adverse effects on several organ systems, including the integumentary, gastrointestinal, neurologic, cardiovascular, endocrine, and respiratory.
Common side effects of hydromorphone include:
- Constipation
- Nausea
- Vomiting
- Dizziness
- Sedation
- respiratory depression
- pruritus
- headache
- Somnolence
- Severe adverse effects of hydromorphone include:
- Hypotension
- Syncope
- adrenal insufficiency
- coma
- raised intracranial pressure.
- seizure
- suicidal thoughts
- apnea
- respiratory depression or arrest
- drug dependence or withdrawal
- neonatal drug withdrawal syndrome
- Hydromorphone is contraindicated in patients with:
- known allergies to the drug, sulfites, or other components of the formulation.
- known hypersensitivity to opioids.
- severe respiratory depression
- paralytic ileus
- acute or severe bronchial asthma (1).
Caution should be exercised in patients with:
- respiratory insufficiency
- head injuries
- increased intracranial pressure.
- liver or kidney impairment.
Considerations for Nurse Practitioners
As nurse practitioners, it is crucial to assess the patient's pain intensity and overall health status before initiating Hydromorphone. Start with the lowest effective dose and titrate carefully for optimal pain control. Regular monitoring for adverse effects, signs of opioid toxicity, and therapeutic response is essential. Educate patients about the potential side effects, proper dosing, and the importance of not exceeding prescribed doses. Additionally, nurse practitioners should be familiar with local regulations and guidelines regarding opioid prescribing and follow appropriate documentation and monitoring practices.
Additional Considerations
In terminal cancer patients, clinicians should not restrain opioid therapy even if signs of respiratory depression become apparent.
Hydromorphone requires careful administration in cases of concurrent psychiatric illness.
Specific Patient Considerations:
- Hepatic impairment and Renal Impairment: Initiate hydromorphone treatment at one-fourth to one-half of the standard starting dosage, depending on the degree of impairment.
- Pregnancy considerations: Hydromorphone can traverse the placental barrier and induce NOWS.
- Breastfeeding considerations: Nonopioid analgesic agents are preferable for breastfeeding women.
- Older patients: hydromorphone is categorized as a potentially inappropriate medication for older adults (1).
Tramadol

Mechanism of Action and Metabolism
Tramadol is a Schedule IV opioid medication with a higher potential for dependency and misuse than non-opioid medications. It binds to opioid receptors in the central nervous system, inhibiting the reuptake of norepinephrine and serotonin. It also has weak mu-opioid receptor agonist activity.
The liver metabolizes tramadol mediated by the cytochrome P450 pathways (particularly CYP2D6) and is mainly excreted through the kidneys.
Tramadol is used for moderate to severe pain.
Available Forms of Tramadol include:
- Immediate-release-typically used for acute pain management.
- Extended-release-used for chronic pain.
Dosing and Monitoring
Tramadol has an oral bioavailability of 68% after a single dose and 90–100% after multiple doses and reaches peak concentrations within 2 hours. Approximately 75% of an oral dose is absorbed, and the half-life of tramadol is 9 hours (18).
Tramadol dosing should be individualized based on the patient's pain severity and response.
The initial dose for adults is usually 50-100 mg orally every 4-6 hours for pain relief. The maximum daily dose is 400 mg for immediate-release formulations and 300 mg for extended-release formulations (18).
It is essential to monitor the patient's pain intensity, response to treatment, and any adverse effects. Regular reassessment and adjustment of the dosage may be necessary.
Side Effects and Contraindications
Tramadol is responsible for severe intoxications leading to consciousness disorder (30%), seizures (15%), agitation (10%), and respiratory depression (5%). The reactions to Tramadol suggest that the decision to prescribe should be carefully considered.
Common Side Effects of Tramadol Include:
- Nausea
- Vomiting
- Dizziness
- Constipation
- Sedation
- Headache
- CNS depression
- Seizure
- Agitation
- Tachycardia
- Hypertension
- reduced appetite
- pruritus and rash
- gastric irritation
Serious side effects include:
- respiratory depression
- serotonin syndrome
- seizures
Contraindications
Tramadol is contraindicated in patients with:
- history of hypersensitivity to opioids
- acute intoxication with alcohol
- opioids, or other psychoactive substances
- Patients who have recently received monoamine oxidase inhibitors (MAOIs)
Additionally, the following can be observed in tramadol intoxication:
- miosis
- respiratory depression
- decreased level of consciousness
- hypertension
- tremor
- irritability
- increased deep tendon reflexes
Poisoning leads to:
- multiple organ failure
- coma
- cardiopulmonary arrest
- death
Considerations for Nurse Practitioners
Tramadol has been increasingly misused with intentional overdoses or intoxications. Suicide attempts were the most common cause of intoxication (52–80%), followed by abuse (18–31%), and unintentional intoxication (1–11%). Chronic tramadol or opioid abuse was reported in 20% of tramadol poisoning cases. Fatal tramadol intoxications are uncommon except when ingested concurrent with depressants, most commonly benzodiazepines and alcohol (18).
Tramadol poisoning can affect multiple organ systems:
- gastrointestinal
- central nervous system: seizure, CNS depression, low-grade coma, anxiety, and over time anoxic brain damage
- Cardiovascular system: palpitation, mild hypertension to life-threatening complications such as cardiopulmonary arrest
- respiratory system
- renal system: renal failure with higher doses of tramadol intoxication
- musculoskeletal system: rhabdomyolysis
- endocrine system: hypoglycemia, serotonin syndrome (18)
Cannabis
Mechanism of Action and Metabolism
Cannabis is classified as a Schedule I status. It contains various cannabinoids, with delta-9-tetrahydrocannabinol (THC) and cannabidiol (CBD) being the most studied. THC primarily acts on cannabinoid receptors in the brain, producing psychoactive effects, while CBD has more diverse effects on the nervous system. These cannabinoids interact with the endocannabinoid system, modulating neurotransmitter release and influencing various physiological processes (32).
Similar to opioids, cannabinoids are synthesized and released in the body by synapses that act on the cannabinoid receptors present in presynaptic endings (32). They perform the following actions related to analgesia:
- Decrease the release of neurotransmitters.
- Activate descending inhibitory pain pathways.
- Reduce postsynaptic sensitivity and alleviate neural inflammation.
- Modulate CB1 receptors within central nociception processing areas and the spinal cord, resulting in analgesic effects.
- Attenuate inflammation by activating CB2 receptors (32).
- Emerging research shows cannabis is indicated for:
- Migraines
- chronic pain
- back pain
- arthritic pain
- pain associated with cancer and surgery.
- neuropathic pain
- diabetic neuropathic pain when administered early in the disease progression.
- sickle cell disease
- cancer
- inflammatory bowel disease (32)
Available Forms
Cannabis refers to products sourced from the Cannabis sativa plant. There are differences between cannabis, cannabinoids, and cannabidiol (CBD). Cannabinoids are extracted from the cannabis plants. Cannabinoid-based treatments, such as dronabinol and CBD, are typically approved medical interventions for specific indications. THC (9-tetrahydrocannabinol) is the psychoactive component of the cannabis plant. CBD is a non-psychoactive component (32).
Cannabis can be consumed in different forms, each with a different onset and duration. Patients may have individual preferences, including:
- smoking/vaporizing dried flowers.
- consuming edibles
- tinctures or oils
- applying topicals (32)
Dosing and Monitoring
Inhaling marijuana via the lungs by smoking or vaping causes maximum plasma concentration within minutes. Psychiatric effects begin within seconds to a few minutes after inhalation and peak after 15 to 30 minutes. The effect diminishes throughout 2 to 3 hours (32).
Oral ingestion of marijuana causes psychiatric effects that typically occur between 30 and 90 minutes and reach maximum effect after 2 to 3 hours. Ingested marijuana effects last about 4 to 12 hours (32).
Dosing cannabis is challenging due to variations in potency and individual responses. Start with low doses and titrate slowly to achieve the desired effect while minimizing side effects. Regular monitoring is crucial, including assessing symptom relief, adverse effects, and potential drug interactions. Encourage patients to keep a diary to track their cannabis use and its effects (32).
Side Effects and Contraindications
Cannabis can exacerbate mental health conditions such as anxiety and psychosis. Common side effects of cannabis include (32):
- Dizziness
- dry mouth
- increased heart rate
- impaired memory
- psychoactive effects
Contraindications include:
- Pregnancy
- Breastfeeding
- heart disease
- respiratory conditions
- history of substance abuse
- mental health disorders

Self Quiz
Ask yourself...
- How do you address patients' misconceptions about pain medications?
- What are the mechanisms of action for commonly prescribed pain medications?
- How do these mechanisms of action contribute to pain relief?
- What are the potential side effects and risks associated with commonly prescribed pain medications?
- How do you educate patients about the risks and benefits of pain medications?
- How do you manage patients who require high-dose opioids for pain management?
- Is medical cannabis legal in your State? If yes, are you familiar with the prescribing guidelines?
- Do you have any personal biases against the use of medical cannabis? Why or why not?
Case Study
Mary is agreeable to trying an increased dose of Gabapentin. Mary would also like to see a counselor to discuss her past and get help with her anxiety. You made an appointment for Mary to see a Licensed Clinical Social Worker in your clinic.
You read the side effects and warnings for Gabapentin, and it is unsafe to use Gabapentin and Tramadol together since they are both depressants. You order a non-steroidal drug for Mary's somatic knee pain and make a consult for imaging studies on her left knee. You also make a referral to Orthopedics.
You educated Mary about the side effects of Gabapentin and scheduled a follow-up appointment. The day after Mary began her treatment with the increased Gabapentin, you called Mary to follow up on its effect. Mary still has pain, but she is not having any untoward side effects. Gabapentin may not work immediately so you will schedule a follow-up call in 3 days.


Self Quiz
Ask yourself...
- In this case study, Mary has insurance. How might your practice be different were Mary not insured?
- In your experience, what are the possible reasons for Mary's knee pain not being a part of her previous treatment record?
- Consider how your assessment of Mary's needs differs from the above-mentioned case study.
- Explain the rationale for decisions made by the nurse practitioner in the case study mentioned above and if your decisions would differ.
Opioid Use, the Opioid Epidemic, and Statistics
The use and misuse of opioids has become a pressing public health concern, leading to a global epidemic. The history of opioid use, the opioid epidemic, and associated statistics provide essential context for healthcare professionals in addressing this public health crisis. More importantly, it is estimated that 1 in 4 patients receiving prescription opioids in primary care settings will misuse them. In addition, 50% of opioid prescriptions are written by primary care providers, including nurse practitioners (22). Understanding the factors contributing to the epidemic and the magnitude of its impact is crucial for effective prevention, intervention, and treatment strategies.
History of Opioid Use
Opioids have a long history of medicinal use, dating back to ancient civilizations. They have been a drug of choice for pain relief for thousands of years. The introduction of synthetic opioids in the 19th century, such as morphine and later heroin, revolutionized pain management. However, their potential for addiction and misuse soon became apparent (16).
The Opioid Epidemic
The opioid epidemic refers to the surge in opioid misuse, addiction, and overdose deaths. The epidemic gained momentum in the late 1990s with increased prescribing of opioids for chronic pain (43).
No doubt, increased prescribing put opioids in the hands of consumers, but increased prescribing resulted from a multifactorial influence. One of the main influences was aggressive marketing by pharmaceutical companies, which has been well publicized. However, due to the long history of underprescribing pain medications for fear of misuse and addiction, the medical community was primed to expand its opioid prescribing practices (31).
A historical event that increased comfort with prescribing opioids, in the writer's opinion, was the introduction of the Medicare Hospice Benefit in 1986. Medical directors must be contracted or employed by hospices, and these medical directors had or soon gained pain management expertise. To further promote hospice and effective pain management, the hospice medical directors, with newly acquired skills, provided education throughout medical communities about pain management and specifically to decrease the fear of using opioids. Pharmacies and attending physicians grew accustomed to giving opioids for home use. Hospice care is for terminally ill patients, defined as a life expectancy of 6 months or less. Still, the reality is that hospice discharges 12 to 40% of patients for ineligibility and other reasons.
A more prominent factor in increasing opioid prescribing was the 1996 American Pain Society's introduction of pain as "the 5th Vital sign." Soon after, The Joint Commission promoted pain as "the 5th Vital Sign" and began compliance surveys in healthcare organizations requiring pain assessment details to be as prominent as blood pressure and heart rate. The Joint Commission cited a quote from 1968 by a nurse from the University of California Los Angeles, Margo McCaffrey, who defined pain as "…Whatever the experiencing person says it is, existing whenever s/he says it does." The Joint Commission accreditation programs pursued pain management as part of the accreditation process throughout its healthcare accreditation programs, including hospice accreditation by 1989 per TJC Timeline (48).
The National Institute of Health published an article about the Joint Commission's role in the opioid epidemic, particularly regarding the definition of pain, "This definition emphasizes that pain is a subjective experience with no objective measures. It also stresses that the patient, not the clinician, is the authority on the pain and that their self-report is the most reliable indicator of pain. This set the tone for clinicians: Patients are always to be trusted to report pain accurately” (45).
Statistics on the Opioid Epidemic
In the United States alone, over 500,000 people died from opioid overdoses between 1999 and 2017. The number of opioid-related overdose deaths continues to increase, with synthetic opioids, mainly illicitly manufactured Fentanyl, playing a significant role in recent years (46). Fentanyl-laced drugs, such as marijuana, are increasingly sold knowing and unknowingly to introduce medications with a high addiction rate, thus creating new consumers. This practice can potentially increase deaths due to the imprecise nature of manufacturing (16).
Opioid-related hospitalizations have also risen substantially. In 2014, there were approximately 1.27 million hospitalizations related to opioids in the United States. These hospitalizations not only place a burden on healthcare systems but also reflect the severe consequences of opioid misuse (3).


Self Quiz
Ask yourself...
- Have you experienced changes to your practice because of the opioid epidemic? If so, what are the changes?
- What is your opinion on the validity of Margo McCaffrey's definition of pain?
- What factors influence your willingness or unwillingness to prescribe opioids?
Federal Regulations on Opioid Prescribing
The history of substance use disorder prevention that promotes opioid recovery and treatment for patients and communities can be traced back to the early 20th century. However, the current approach to addressing opioid addiction and promoting healing has evolved significantly in recent times (36).
In the early 1900s, health professionals treated opioid addiction with punitive measures, including incarceration and moralistic approaches. The focus was on punishing individuals rather than providing effective treatment. This approach persisted for several decades until the mid-20th century when the medical community started recognizing addiction as a medical condition rather than a moral failing (36).
The Controlled Substances Act (CSA), introduced in 1970, was a response to increasing drug abuse and illicit drug trafficking in the United States. The CSA is a federal law regulating the manufacture, possession, distribution, and use of certain substances, including drugs and medications, that can potentially cause abuse and dependence. Its primary purpose is to combat drug abuse, reduce drug-related crimes, and protect public health and safety. The Drug Enforcement Agency (DEA) plays a crucial role in enforcing the CSA by monitoring and controlling controlled substance production, distribution, and use (31).
In the 1990s, the significant increase in opioid prescribing, leading to a surge in opioid addiction and overdose deaths, prompted a shift in focus toward prevention. Efforts were made to educate healthcare providers about the risks of overprescribing opioids and to implement prescription drug monitoring programs to track and prevent abuse (36).
The Comprehensive Addiction and Recovery Act (CARA) was signed into law in 2016 to expand access to treatment and recovery services for opioid addiction. This legislation allocated funding for prevention, treatment, recovery, and support services while promoting evidence-based practices and programs (36).
The Centers for Disease Control and Prevention (CDC) published guidelines in 2016 for prescribing opioids for chronic pain, which was updated in 2022. These guidelines emphasize the importance of non-opioid alternatives, using the lowest effective dose for the shortest duration, and assessing the benefits and risks of continued opioid therapy (13).
Furthermore, the Substance Use Disorder Prevention that Promotes Opioid Recovery and Treatment for Patients and Communities Act (SUPPORT) was signed into law in 2018, providing additional resources to address the opioid crisis. This legislation expanded access to medication-assisted treatment (MAT), increased the availability of naloxone, a medication used to reverse opioid overdose, and enhanced support for recovery housing (36).
In recent years, there has been a growing recognition of the importance of a comprehensive approach to opioid addiction, including harm reduction strategies, increased access to naloxone, and the integration of mental health services. Communities and organizations have been working together to address the underlying issues contributing to addiction, such as poverty, trauma, and social determinants of health (50).
Overall, the history of substance use disorder prevention that promotes opioid recovery and treatment has evolved from a punitive approach to a more compassionate and evidence-based model. Efforts are now focused on prevention, early intervention, and expanding access to comprehensive treatment and support services for individuals and communities affected by opioid addiction (36).
The most current federal regulations on opioid prescribing for healthcare providers are the amendments to the CSA in 2018, which added new rules to limit the quantity and duration of opioid prescriptions for acute pain to seven days. In 2022, the CDC updated recommendations to the Clinical Practice Guidelines for Prescribing Opioids for Pain.
The 2022 CDC guidelines are summarized below (13):
- Non-opioid therapies should be considered the first-line treatment for chronic pain.
- Establish clear treatment goals with patients, including realistic pain management and functional improvement expectations.
- Conduct a thorough risk assessment for potential harms before initiating opioid therapy.
- When opioids are used, start with the lowest effective dose and consider immediate-release opioids instead of extended-release or long-acting opioids.
- Prescribe the lowest effective dose for the shortest duration possible, typically three days or less and rarely exceeding seven days.
- Reassess benefits and risks within one day after prescribing opioids, including checking the prescription drug monitoring database.
- Avoid prescribing opioids and benzodiazepines concurrently whenever possible due to the increased risk of overdose and death.
- Offer naloxone to patients at increased risk of opioid overdose, including those with a history of overdose, substance use disorder, or concurrent benzodiazepine use.
- When opioids are no longer needed, taper the dose gradually to minimize withdrawal symptoms.
- Arrange an evidence-based treatment for patients with opioid use disorder, including medication-assisted treatment (Naltrexone, Buprenorphine, or Methadone).

Self Quiz
Ask yourself...
- What are the guidelines general for prescribing opioids for acute pain?
- How do these guidelines differ for chronic pain management?
- Discuss how federal regulations impact the practice of nurse practitioners in terms of opioid prescribing.
- Describe the potential benefits and challenges nurse practitioners face when adhering to federal regulations on opioid prescribing.
- How can nurse practitioners navigate and stay updated with evolving federal regulations surrounding opioid prescribing to ensure safe and effective care?
- How do you ensure appropriate documentation when prescribing controlled substances?
Safe Prescribing and Prescription Monitoring Program
Prescription Drug Monitoring Programs (PDMP) are state-run electronic databases that track.
the prescribing and dispensing of controlled substances. PDMPs are designed to improve patients.
care and safety by giving clinicians access to patients' prescription histories, allowing them to make informed decisions when prescribing controlled substances. PDMPs help identify patients at risk of substance misuse or prescription drug overdose. They also enable clinicians to identify potential drug interactions and prevent opioid diversion (14).
PDMPs collect and store data from pharmacies and prescribers in a centralized database. Clinicians can access this database to review a patient's prescription history, including the types of medications prescribed, the prescribers involved, and the dispensing pharmacies (14).
In many states, PDMP use is mandated by law, and nurse practitioners may be required to register and use the system. It is essential to understand state-specific laws and regulations regarding PDMP use.
PDMPs have some limitations, such as incomplete data or delays in reporting. The CDC emphasizes that clinicians should use PDMP data for their clinical assessment and other relevant information to make informed decisions about prescribing controlled substances. Still, PDMP cannot be used as the sole basis for denying or providing treatment (14).
Case Study
After five days on Gabapentin, Mary was doing well, and her neuropathic pain had decreased to 3/10. However, Mary suffered a fall after her knee "gave out" and injured her knee and back. She was in severe pain, and her family drove her to the ER. The ER doctors saw Mary, and orthopedics were consulted. Mary has surgery scheduled for a knee replacement a week from now.
Mary was prescribed Vicodin because she was in excruciating pain, but her prescription only allowed enough medication for two days. Mary has made an appointment with you to renew her prescription.
You evaluate Mary because you know that concomitant use of Gabapentin and opioids puts Mary at risk for respiratory depression and possible side effects, including accidental overdose.
Mary stated she has been more alert the past 24 hours and is afraid her functional status will continue to decline if she does not have more Vicodin because the pain in her back and knee makes it difficult to stand. You assess Mary. Mary stated she occasionally drinks alcohol but has not had a drink since she moved. She has no familial history of substance abuse or mental health disorders.
Mary's mother stayed at her house to help her for the first 24 hours after Mary's return from the ER, but Mary is providing her care now.
You check the PDMP database and see that Mary was prescribed eight pills she has taken over the last 48 hours.
Since the Vicodin has been effective without untoward side effects, and Mary's function is improving, you decide to refill the prescription of Vicodin. You will taper the dose to three Vicodin daily for two days and two for one day. Mary will be near her appointment for a knee replacement as well.

Self Quiz
Ask yourself...
- What are the potential benefits and drawbacks of using PDMPs in your practice?
- How can PDMPs help you identify potential drug abuse or diversion cases among your patients? Can you provide examples from your own experience?
- In what ways do PDMPs impact your decision-making process when prescribing controlled substances?
- What are the key considerations when prescribing controlled substances?
- How do you ensure responsible prescribing practices for controlled substances?
Preventing Opioid Use Disorder
As previously discussed, opioid addiction is a growing concern worldwide, affecting individuals from all walks of life. According to the CDC, "Anyone who takes prescription opioids can become addicted to them" (14).
As frontline healthcare professionals, nurse practitioners must recognize the signs of opioid addiction to provide timely intervention and support. This section will outline the key indicators of opioid addiction.
Physical Symptoms
Physical symptoms are often the first noticeable signs of opioid addiction. These symptoms may include constricted pupils, drowsiness, slurred speech, impaired coordination, and increased sensitivity to pain. Additionally, individuals struggling with opioid addiction may exhibit frequent flu-like symptoms, such as a runny nose, sweating, itching, or gastrointestinal issues.
Behavioral Changes
Opioid addiction can significantly impact an individual's behavior. These may include increased secrecy, frequent requests for early prescription refills, doctor shopping (seeking prescriptions from multiple healthcare providers), neglecting personal hygiene, and experiencing financial difficulties due to excessive spending on opioids (37).

Social Isolation
Opioid addiction often leads to social withdrawal and isolation. Individuals struggling with opioid addiction may distance themselves from family, friends, and social activities they once enjoyed. They may exhibit erratic mood swings, become defensive or hostile when confronted about their drug use, and display a general lack of interest in previously important activities (30).
Psychological Changes
The psychological impact of opioid addiction is significant. Individuals with opioid addiction may exhibit increased anxiety, depression, irritability, and restlessness. They may also experience cognitive impairments, memory lapses, and difficulties in decision-making. Healthcare professionals should be attentive to these changes, as they can indicate opioid addiction (51).
Tolerance and Withdrawal Symptoms
The development of tolerance and withdrawal symptoms are critical signs of opioid addiction. Individuals may require increased dosages of opioids to achieve the desired effect, indicating a growing tolerance. Furthermore, withdrawal symptoms such as muscle aches, nausea, vomiting, insomnia, and intense cravings for opioids may occur when the drug is discontinued or reduced abruptly (51).

Self Quiz
Ask yourself...
- Discuss how nurse practitioners can contribute to preventing opioid use disorder.
- Explain how nurse practitioners effectively communicate the risks and signs of opioid misuse without stigmatizing or alienating patients.
- What are the signs of opioid addiction or misuse in patients?
- How do you approach patients who may be at risk for opioid addiction?
- How do you ensure appropriate documentation when prescribing controlled substances?
Opioid Overdose
The management of opioid overdose, withdrawal, and addiction requires a comprehensive approach that combines pharmacological interventions with psychosocial support. Naloxone remains a vital tool for reversing opioid overdose, while medications such as Methadone, buprenorphine, and naltrexone play crucial roles in withdrawal and addiction treatment (National Institute of Health, 2023). Nurse practitioners must stay vigilant and informed about the evolving landscape of medications. This section aims to provide a comprehensive review of medications and treatment strategies for opioid overdose, withdrawal, and addiction and is excerpted from the NIH (40).
Naloxone
Mechanism of Action and Metabolism
Naloxone is an opioid receptor antagonist. It works by binding to opioid receptors and displacing any opioids present, thereby reversing the effects of opioid overdose. It has a higher affinity for opioid receptors than most opioids, effectively blocking their action.
Naloxone is indicated for emergency intervention of opioid overdose. It effectively reverses respiratory depression and other life-threatening effects. Studies suggest the potential benefits of combining naloxone with other medications, such as buprenorphine (see below), to improve outcomes. Initiatives promoting community-based naloxone distribution programs have shown promising results in reducing opioid-related deaths.
Available Forms
Naloxone is available in various formulations:
- Intranasal
- Intramuscular
- Intravenous
- auto-injectors.
The most used form is the intranasal spray, which is easy to administer and requires no specialized training. Intranasal naloxone formulations have gained popularity due to their ease of use and increased availability. A recent study showed that the non-FDA-approved compound spray was far less effective than either FDA compound (15).
Dosing and Monitoring
The recommended initial dose of naloxone for opioid overdose is 2mg intranasally or 0.4mg to 2mg intramuscularly or intravenously. If the patient does not respond within 23- minutes, additional doses may be administered every 2-3 minutes. Continuous monitoring of the patient's respiratory status is essential, as repeat doses may be required due to the short half-life of naloxone.
Side Effects and Contraindications
Naloxone has been shown not to affect individuals without opioids in their system.
Common side effects of naloxone include
- Withdrawal symptoms: increased heart rate, sweating, and agitation
- nausea
- vomiting
- headache
Contraindications include known hypersensitivity to naloxone and situations where the use of naloxone may be unsafe or not feasible.
Considerations for Nurse Practitioners
Fentanyl and other opioids have a rapid onset, and the need to act quickly is paramount. As mentioned previously, the ease of use and higher plasma concentrations using the FDA-approved 4-mg FDANxSpray device compared with the locally compounded nasal sprays should be considered when ordering Naloxone (15).
Fentanyl and other potent synthetic opioids may require multiple administrations of naloxone to achieve reversal of an overdose (Chiang, Gyaw, & Krieter, 2019). As a nurse practitioner prescribing naloxone, it is crucial to assess the patient's risk factors for opioid overdose, such as a history of substance use disorder or chronic pain management. Education regarding the proper administration of naloxone should be provided to the patients and their caregivers. Additionally, it is essential to provide resources for follow-up care, including addiction treatment and ongoing support.
Methadone
Mechanism of Action and Metabolism
Methadone is a long-acting opioid agonist that effectively suppresses withdrawal symptoms and reduces cravings. It binds to the same opioid receptors in the brain as other opioids. It relieves withdrawal symptoms and reduces cravings by blocking the euphoric effects of opioids, thus helping individuals with opioid dependence to achieve stability (33).
Available Forms
Methadone is available in oral tablets and liquid formulations. The oral tablet is the most used form and is typically administered once daily (33).
Dosing and Monitoring
Methadone dosing is individualized based on the patient's response and needs. Initially, the dose often started low and gradually increased until the patient reached a stable dose. Dosing may need to be adjusted based on the patient's response, adherence, and any changes in their overall health. Regularly monitoring the patient's vital signs, urine drug screens, and assessment of their withdrawal symptoms and cravings is essential.
Side Effects and Contraindications
Common side effects of methadone include:
- Constipation
- dry mouth
- drowsiness
- sweating
- weight gain
- respiratory depression
Contraindications include:
- known hypersensitivity to methadone
- severe asthma
- respiratory depression
- certain heart conditions (33).
Considerations for Nurse Practitioners
As a nurse practitioner prescribing methadone, conducting a comprehensive assessment of the patient's medical history, current medications, and substance use history is crucial. Opioid treatment programs or specialized clinics are often involved in methadone treatment, so collaboration and coordination of care with these programs are essential. Regularly monitoring the patient's progress, adherence, and potential side effects or drug interactions is essential. Additionally, providing education on the risks and benefits of methadone and the importance of adherence to the prescribed regimen is crucial for successful treatment outcomes.
Buprenorphine
Mechanism of Action and Metabolism
Buprenorphine is a partial opioid agonist with a ceiling effect that minimizes the risk of overdose while reducing withdrawal symptoms. Buprenorphine is a partial opioid agonist that binds to the same receptors as other opioids but produces a weaker response. It has a high affinity for the mu-opioid receptors, which helps reduce cravings and withdrawal symptoms in individuals with opioid dependence.
Available Forms
Buprenorphine is available in different formulations, including sublingual tablets, buccal films, and extended-release injections. The sublingual tablets have different strengths, such as 2mg, 4mg, 8mg, and 12mg. Buprenorphine is taken as a daily tablet or weekly or monthly injection.
Dosing and Monitoring
The dosing of buprenorphine varies depending on the individual's opioid dependence severity and treatment phase. Initially, a low dose (e.g., 2-4mg) is given, and it may gradually increase to a maintenance dose of 8-24 mg daily. Regular monitoring is essential to assess the patient's response, adherence, and potential side effects.
Side Effects and Contraindications
Common side effects of buprenorphine include:
- Constipation
- Nausea
- Headache
- Insomnia
- Sweating
Serious side effects are rare but can include:
- Respiratory depression
- Allergic reactions
Buprenorphine is contraindicated in individuals with:
- Severe respiratory insufficiency
- Acute intoxication with opioids
- Known hypersensitivity
Considerations for Nurse Practitioners
Nurse practitioners can prescribe buprenorphine for opioid dependence treatment under the Drug Addiction Treatment Act (DATA). To become eligible, they must complete specific training requirements and obtain a waiver from the Substance Abuse and Mental Health Services Administration (SAMHSA). Nurse practitioners should assess patients thoroughly, including their opioid use history, comorbidities, and medication compatibility, while ensuring appropriate counseling and referral for comprehensive treatment (40).
Clonidine + Lofexidine
Mechanism of Action and Metabolism:
Both Clonidine and Lofexidine are alpha-2 adrenergic agonists. They work by stimulating alpha-2 receptors in the brain, which reduces sympathetic outflow and norepinephrine release. This results in decreased sympathetic activity, leading to various effects such as reduced blood pressure, decreased heart rate, and alleviated withdrawal symptoms (28).
Available Forms
Clonidine is available in oral tablets and patches. Lofexidine is available in oral tablets and is taken as needed (40).
Dosing and Monitoring
For opioid withdrawal, the Clonidine dose ranges from 0.1-0.3 mg every 4-6 hours. Lofexidine is usually initiated at 0.53 mg three times daily, and the dose can be increased to 2.88 mg daily. Monitoring blood pressure and heart rate is essential during treatment (40).
Side Effects and Contraindications:
Common side effects of both medications include:
- dry mouth
- sedation
- dizziness
- constipation
- orthostatic hypotension (40).
Both medications are contraindicated in patients with:
- Hypotension
- Bradycardia
- heart block
- history of hypersensitivity to the drugs (40).
Considerations for Nurse Practitioners:
An early study of lofexidine vs. clonidine for withdrawal symptoms showed that treatment with lofexidine resulted in lower withdrawal symptoms, fewer mood problems, less sedation, and hypotension. There were no significant differences in craving levels, morphine metabolites in urine, or dropout rates when both were compared.
Lofexidine can be a safe option for outpatient treatment as it does not lead to hypotension. However, nurse practitioners must closely monitor patients' blood pressure and heart rate during treatment and educate them about possible side effects. If patients experience any concerning symptoms, they should inform their nurse practitioner immediately.
Gradual dose reduction of Clonidine is crucial to prevent rebound hypertension. Before prescribing either medication, nurse practitioners should assess for any contraindications or potential drug interactions (19).
Emerging Therapies for Withdrawal
Extended-release naltrexone: Naltrexone is an opioid receptor antagonist that blocks the effects of opioids, reducing the risk of relapse. It is taken as a monthly injection.
Alpha-2 adrenergic agonists: Emerging evidence suggests the potential use of dexmedetomidine and guanfacine for managing opioid withdrawal symptoms.
Medication-Assisted Treatment (MAT):
Methadone was introduced in the 1960s and marked a significant turning point in opioid addiction treatment or MAT. Along with counseling and behavioral therapies, MAT became the cornerstone of opioid addiction recovery.
Examples of medications used:
- Methadone
- Buprenorphine:
- Naltrexone:
Adjunctive Pharmacotherapies:
Antidepressants: Selective serotonin reuptake inhibitors and tricyclic antidepressants may help manage co-occurring depression and anxiety.
Anticonvulsants:
Medications like Gabapentin and pregabalin show promise in reducing opioid cravings and improving treatment outcomes.


Self Quiz
Ask yourself...
- What are the mechanisms of action for commonly prescribed addiction medications?
- What are the potential risks and benefits of using benzodiazepines for pain management?
- How do you assess and manage patients with co-occurring pain and substance use disorders?
- What are the guidelines for prescribing addiction medications like buprenorphine or methadone?
- How do these medications work in the treatment of opioid use disorder?
- What are the potential side effects and risks associated with addiction medications?
- How do you support patients in their recovery from opioid use disorder?
- How do you address patients' concerns and fears about addiction medications?
- What are the federal guidelines around prescribing addiction medications for nurse practitioners?
- How do these guidelines influence your prescribing practices?
Other Substance Use Disorders
Patients in pain may struggle with Substance Use Disorders other than Opioid Use Disorder. Substance use disorders may often occur with mental health conditions such as anxiety, depression, and bipolar disorder. In addition, many individuals engage in polydrug use. Understanding the most common Substance Use Disorders aids in a comprehensive assessment of the patient and the development of appropriate treatment plans (28).
Alcohol Use Disorder (AUD):
The prevalence of AUD worldwide was estimated to be 9.8% in men and 5.5% in women in 2016 (28).
Cannabis Use Disorder (CUD):
the prevalence of CUD in the United States increased from 2.18% in 2001-2002 to 2.89% in 2012-2013. (28).
Cocaine Use Disorder:
According to the National Survey on Drug Use and Health (NSDUH), in 2019, approximately 1.9 million Americans aged 12 or older had cocaine use disorder in the past year (44).
Methamphetamine Use Disorder:
A study published in Drug and Alcohol Dependence reported that the prevalence of methamphetamine use disorder in the United States was estimated to be 0.2% in 2015-2016 (6).

Self Quiz
Ask yourself...
- What are the options available for managing opioid addiction and withdrawal?
- How can nurse practitioners support patients in their recovery from opioid addiction?
- What strategies can nurse practitioners employ to effectively engage and build trust with patients reluctant to disclose or seek help for substance abuse disorders?
- How can nurse practitioners collaborate with other healthcare professionals and community resources to provide comprehensive care and support for patients with substance abuse disorders?
- What techniques or tools can nurse practitioners employ to start these sensitive conversations with new patients?
- How do you assess and manage patients experiencing opioid withdrawal symptoms?
- What are the non-pharmacological interventions for managing opioid withdrawal?
- How do you educate patients about the risks and benefits of addiction medications?
- How do you monitor patients on addiction medications for adherence and progress?
- What are the drug potential interactions with commonly prescribed addiction medications?
Drug Diversion and Illegal opioids
Misuse of opioids is facilitated by diversion and is defined as "the transfer of drugs from lawful to unlawful use" (24). Most commonly, this occurs when family and friends share prescribed opioids with other family and friends. Opioids and other controlled drugs are also diverted from healthcare facilities. Statistics show that healthcare facility diversion has increased since 2015 (24)
Diversion affects patients, healthcare workers, healthcare facilities, and public health. Patients experience substandard care due to ineffective pain management and impaired healthcare workers. In addition, affected patients are at risk of infections from compromised syringes (24).
Healthcare employees who divert are at risk of overdose and death. If caught, they face criminal prosecution and malpractice suits. Healthcare facilities also bear the cost of diverted drugs via internal investigations, follow-up care for affected patients, regulatory fines for inadequate safeguards, and declining public trust (24).
Despite the enormous consequences of drug diversion, healthcare facilities have implemented few processes to detect and deter the diversion of controlled substances (24).

Self Quiz
Ask yourself...
- What protocols can nurse practitioners implement to prevent drug diversion within their healthcare setting?
Patient Teachings and Considerations
Opioids have significant side effects and carry a risk of addiction and overdose. Nurse practitioners can decrease the risks of misuse and addiction by educating patients on appropriate disposal, safe storage, and potential signs of addiction. Taking additional time to provide teaching nurse practitioners can promote patient safety, informed decision-making, and responsible opioid use.
Safe Storage and Disposal:
- Teach patients to store opioids securely, out of reach of children, pets, visitors, and non-caregiver family members, to prevent accidental ingestion or misuse (13). Only the caregiver, if applicable, or the patient should have access to pain medications.
- Instruct patients on proper disposal methods, such as using drug take-back programs or mixing opioids with undesirable substances (e.g., coffee grounds) before throwing them away (11) (13).
Medication Adherence:
- Emphasize the importance of taking opioids as prescribed, at the correct dose and frequency, to achieve optimal pain relief.
- Encourage patients to notify their healthcare provider if they experience inadequate pain control or side effects (35).
Potential Side Effects:
- Educate patients about common side effects of opioids, including constipation, nausea, sedation, and respiratory depression.
- Discuss strategies to manage side effects, such as maintaining adequate hydration, consuming a fiber-rich diet, and using over-the-counter laxatives as needed (11).
Risk of Dependence and Addiction:
- Explain the potential for opioid dependence and addiction, especially with long-term use or a history of substance abuse.
- Encourage patients to promptly report signs of opioid misuse, such as craving, loss of control, or continued use despite negative consequences (51).

Avoiding Alcohol and Other Central Nervous System Depressants:
- Instruct patients to avoid consuming alcohol or other medications that can enhance the sedative effects of opioids, increasing the risk of respiratory depression.
- Advise patients to contact the Nurse Practitioner before starting new medications, including over-the-counter drugs or herbal supplements (2).
Driving and Operating Machinery:
- Inform patients about the potential impairment caused by opioids, including reduced alertness, reaction time, and coordination.
- Advise patients to avoid driving or operating heavy machinery while taking opioids until they know how the medication affects them (14).

Self Quiz
Ask yourself...
- What strategies can nurse practitioners employ to effectively communicate the risks and benefits of opioid use while ensuring they clearly understand the potential side effects and the importance of adhering to the prescribed regimen?
- How can nurse practitioners promote patient engagement and shared decision-making regarding opioid pain management, considering the potential for dependence and addiction?
- How can nurse practitioners assess a patient's knowledge and understand the safe storage and disposal of opioids?
Case Study
You take some extra time with Mary to educate her on the taper dose of Vicodin, the potential for harm, and the risk of opioids, especially when used concomitantly with Gabapentin. You let Mary know it is unsafe to use alcohol, not only with Vicodin but also with Gabapentin. You let Mary know that Vicodin has a risk of dependency and misuse and, therefore, she will be monitored carefully. You also educate that Mary should store the Vicodin away from visibility by anyone but herself since she can self-administer her medication. You let Mary know that Vicodin can cause constipation and that she should increase her water intake and take a stool softener.
You ask Mary to call you if her pain is not adequately relieved or if her medications run out before the three days.
You let Mary know that if she does stop taking the Vicodin before she has completed all the medication, she should dispose of it by mixing the pills with liquid and coffee grounds to make them unpalatable to animals and others.
Mary complied with your education, completed her course of Vicodin, and was scheduled for surgery. Mary's social worker helped her communicate with her new employer and delayed her start date until after her recovery.
During her recovery, Mary received physical therapy and a short course of pain medication managed by her orthopedist.
Mary returned to the clinic for a follow-up visit after completing her therapy and before starting work. Mary's pain level in her knee is 3/10, and she already feels like she can walk further than pre-surgery. Gabapentin has continued to help Mary's neuropathic pain in her back, and she reports 2/10. Mary looks forward to beginning her new job and is optimistic about the future.
Conclusion
Pain management is the leading cause of primary care appointments and chronic pain is the leading cause of disability. Yet, prescribing opioids for primary care patients is also a factor in drug misuse and the opioid epidemic. Nurse practitioners are challenged to appropriately treat pain and effectively control diversion, addiction, and death from overdose.
It is imperative that nurse practitioners use evidence-based practices to assess, appropriately intervene, and educate about the benefits and potential harm caused by treatment with opioids. Nurse practitioners must stay up to date with the current federal regulations regarding PDMPs, clinical prescribing guidelines, and emerging treatments for pain and opioid abuse disorders.
Tirzepatide for Type 2 Diabetes and Weight Management
Introduction
The emergence of the drug tirzepatide is becoming more popular and widespread and is being utilized among those with diabetes and also those who desire to lose weight. It is one of the newest diabetic drugs given by injection that also triggers dramatic weight loss in those who use the injections.
The U.S. Food and Drug Administration (FDA) approved tirzepatide in 2022 for individuals with diabetes, particularly Type 2 Diabetes. The FDA officials have not approved tirzepatide yet for weight loss, but they are currently tracking the medication and may have a recommendation for its approval by the end of this year. Clinical trials have shown that individuals with an elevated body mass index (BMI) and who did not have diabetes lost a considerable amount of weight when they received tirzepatide (1).
Advanced Practice Registered Nurses (APRNs) need to understand how to safely prescribe tirzepatide and the reasoning as to why it causes weight loss for specific individuals.
Drug Classification
Tirzepatide is part of a class of medications called glucose-dependent insulin tropic polypeptide (GIP) receptor and glucagon-like peptide-1 (GLP-1) receptor agonists. It comprises a 39 amino acid linear synthetic peptide conjugate to selective receptor agonists in preclinical and clinical trials.
Tirzepatide is used for treating Type II diabetes in adults as an adjunct to diet and exercise. It is also used for weight loss in some individuals and has gained increased attention as a new therapeutic agent for glycemic and weight control.
Social media has had a significant influence and increased the desire to use tirzepatide, and while individual results vary, the weight loss in adults ranged from 12 – 25 pounds.
Online pharmacies, diet clinics, and medical spas are implementing thousands of ads on social media to capitalize on a surge of interest in the drug.

Self-Quiz
Ask Yourself...
- Why has there seemed to be an increase in patients requesting this medication? What other medicines intended for type 2 diabetes are also being used for weight loss management?
- What are the ethical considerations regarding marketing this drug for weight loss when its primary use is for type 2 diabetes? Could this impact supply and costs?
Indications of Usage
The use of tirzepatide is being used for both Type II diabetes and weight control in certain patients. It has been a game changer for people living with Type II diabetes. The drug’s primary use is as an adjunct to diet and exercise to improve glycemic control in adults with diabetes.
The drug has also proven beneficial for weight loss in patients experiencing obesity, and those who are taking the highest dosage have shared a body weight reduction of 15.7% (2). Tirzepatide is an injectable prescription medication used together with diet and exercise, and it is not yet known if it can be used safely with patients who have had pancreatitis.
It is important to remember that it is not to be used for patients with Type I diabetes, but it is safe for Type II diabetic patients. Also, the safety of tirzepatide has yet to be discovered for children and those under 18; therefore, the medication should not be used for this age group.
In studies conducted with or without diabetic medicines, 75% – 90% of patients taking tirzepatide reached an overall A1C of less than 7% with an average starting A1C of 7.9 – 8.6% across the following dosages – 5mg, 10mg, and 15mg. The study results were measured at weeks 40 and 52 (3).

Self-Quiz
Ask Yourself...
- What dietary and activity recommendations can you provide to patients using tirzepatide for weight loss?
- Is this drug intended for those who want to lose 5-10 pounds?
Use of Tirzepatide with Diabetic Patients
Tirzepatide can be used for patients with Type II diabetes in combination with a diabetic-friendly diet and exercise. The drug works by lowering the patient’s overall blood sugar and also improves the A1C results of patients over some time. The injection has been approved by the FDA to treat Type II diabetes and is administered once weekly (4).
It is considered the first in a new class of medications – a dual glucose-dependent insulin tropic polypeptide (GIP) and glucagon-like-peptide-1 (GLP-1) receptor antagonist. The mechanism of how it works mimics two gut hormones (GIP and GLP-1). These hormones are essential in how patients digest food and regulate blood glucose after meals. The hormones also play a role in making individuals feel fuller and curb specific food cravings.
The provider can prescribe tirzepatide before attempting other diabetic medications if a patient has a BMI of 30 or greater or 27 or greater with weight-related conditions and if the drug is combined with a personalized weight loss plan that addresses physical activity, nutrition, and lifestyle changes.
However, due to the cost and some insurance companies not covering the injection unless the patient has both diabetes and obesity, the provider must carefully consider prescribing this medication.

Case Study
The patient states this ‘miracle drug’ is worth paying for out of pocket!
Jeff Capron, a 53-year-old Boonville, New York, web developer, started taking tirzepatide in December 2022. His friend had reported good results with the medication, so Jeff looked into the research studies behind it and then spoke with his primary physician.
The physician said, “Yeah, let’s give it a shot,” even though he did not have much experience with it. The physician did not have an opinion one way or the other than looking at the data set and seeing no reason why they could not try it.
Jeff’s hemoglobin A1C went from 10.1% to 6% in 3 months, which was very promising. “I never had that kind of experience with any medication for diabetes.” There is a range in how much A1C reduction people experience with tirzepatide, but many people taking it can get their A1C under 7% — an ideal goal for people with Type 2 diabetes.
Jeff experienced constipation and a little trouble sleeping early, but both issues disappeared quickly. He says, “I wake up in the morning, and my fasting blood sugars are normal.”
The medication took effect, he says, within 12 hours. He compared the feeling to having a gastric bypass.
“You cannot overeat food. As soon as you overeat, you almost feel ill.” While it generally takes a few months to notice effects like A1C reduction and significant weight loss, side effects such as lower appetite may be felt immediately.
Weight loss was not his primary goal, but he lost about 35 pounds on the medication in the first five months. He also lost his sweet tooth. “I can maybe count three sweet things I have eaten since December.”
Jeff found that his appetite slowly recovered days after taking tirzepatide. “You take the shot every Sunday, and by Saturday, you start to get a lot of appetite,” he says. “It does not seem to affect your weight. If I eat a little bit more on Saturday night, on Sunday, the scale will not move one way or the other.”
Jeff is allergic to hornets, so he already carries an auto-injector. He was not worried about using another drug delivered through a needle. “It’s just a push button,” he says. It also helped that his wife is a nurse. “So, I had her with me the first time to ensure I was doing it right. I didn’t even feel it.”
When Jeff was first prescribed tirzepatide, his insurance covered it. The company has since removed that benefit. He has filed an appeal but pays about $1,000 monthly out of pocket for his weekly injections. He plans to keep paying as long as necessary.
He considers the financial burden well worth it. “I have never had a medication that worked as well before for chronic conditions,” Jeff says. “I’ve been blown away by it. For me, it’s a miracle drug. It got rid of my diabetes” (4).

Self Quiz
Ask yourself…
- Can a provider willfully choose to prescribe tirzepatide before other diabetic medications are attempted?
- Would that impact his insurance coverage if Jeff did not meet the clinical criteria for using tirzepatide?
Use of Tirzepatide for Weight Loss Management
As mentioned, this medication is indicated for patients with a BMI of >30 or a BMI of >27 with qualifying comorbidities. Obesity can become a chronic lifetime disease, and for conditions such as these, the patient needs to implement therapy for the lifetime of the disease.
In a study conducted for tirzepatide, there was a dramatic increase in effectiveness compared to traditional nonsurgical interventions such as diet, exercise, and lifestyle changes. However, it has been noted that taking tirzepatide on an ongoing basis is recommended and necessary to maintain any weight loss achieved from the medication.
If a patient stops taking the drug, likely, it will no longer work (5).
Public health officials have expressed concerns about using the drug long-term. Still, data is currently lacking regarding long-term effectiveness, treatment duration, and maintaining weight reduction once the therapy is discontinued.
A recent trial consisted of 783 participants with a BMI greater than 30, and these participants agreed to take either a 10mg or 15mg dose of tirzepatide over 36 weeks. The injection is given once weekly, so this would equal a total of 36 injections.
By the end of 36 weeks, participants lost more than 21% of their body weight. After 36 weeks, participants continued on tirzepatide or received placebo treatment for the following year. The patients needed to be made aware of which treatment they were receiving.
Those still taking tirzepatide injections weekly after 88 weeks lost an additional 7% of their body weight, and those taking the placebo regained 15% at the end of 88 weeks (5).

Self Quiz
Ask yourself…
- What is the minimum BMI needed to qualify to receive this drug for weight loss management?
- Is this medication indicated for long-term use for patients with a high BMI?
Common Side Effects and Contraindications
Side Effects
Patients vary immensely with different experiences and side effects related to tirzepatide; however, the following are the most common side effects experienced by those taking the medication:
- Nausea
- Decreased appetite
- Vomiting
- Diarrhea
- Indigestion
- Constipation
- Stomach Pain
Tirzepatide usually does not cause fatigue, leaving one feeling weak, tired, and low energy. However, fatigue can be a common side effect of Type II diabetes.
It is important to note that most individuals who experience nausea, vomiting, and diarrhea episodes do so while the dosage increases, and typically, the symptoms decrease over time. G.I. effects were more prominent in those taking tirzepatide than those taking the placebo. The individuals not in the placebo group were more likely to stop treatment due to the unpleasant side effects (3).


Self Quiz
Ask yourself...
- Does tirzepatide cause fatigue in patients who use it?
Contraindications
Tirzepatide may cause thyroid tumors, including thyroid cancer, and it is essential to watch for possible symptoms, such as swelling or a lump in the neck, hoarseness, shortness of breath, or trouble swallowing.
Tirzepatide should also not be prescribed to any patient with Type 1 Diabetes.
One of the main ways that tirzepatide works is by stimulating the release of insulin from the pancreas, and due to this fact, there have not been many studies and clinical trials that include those with Type I diabetes.
However, this is not to say that prescribers have never ordered tirzepatide for those with Type I diabetes. Still, it is essential to note that if prescribed, it would be in addition to traditional insulin therapy.
- Personal or family history of a type of thyroid cancer known as medullary thyroid carcinoma (MTC).
- Any history of Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
- Patients who are allergic to the actual medication or any of its ingredients.
- Younger than 18 years of age


Self Quiz
Ask yourself...
- What is the reason that tripeptide is contraindicated in those with Type I diabetes?
- Why is there a risk with patients who have a thyroid disorder?
Safe Prescribing Practices, Guidelines, and Considerations for Providers
Safe Prescribing Practices
As with all prescribed medications, safe standards of care must be implemented and followed to ensure patient safety is maintained. The same applies to providers considering prescribing tirzepatide, and specific criteria must be met beforehand. The following information discusses guidelines involving exclusion and inclusion criteria for providers to prescribe tirzepatide (6) accurately.
Guidelines
Exclusion Criteria – If present, the following indicates that the patient should not receive tirzepatide:
- Diagnosis of Type I diabetes
- Personal or family history of medullary thyroid carcinoma or with Multiple Endocrine Neoplasia syndrome type 2
- Severe gastrointestinal dysmotility
- History of pancreatitis
- Pregnancy
- Proliferative Diabetic Retinopathy (PDR), severe Nonproliferative Diabetic Retinopathy (NDR), clinically significant myalgic encephalomyelitis (M.E.), or diabetic macular edema (DME) unless the risks/benefits have been discussed with the patient and are documented in the patient's health record along with monitoring plans and follow-up with an eye specialist who is informed at the time of initiation.
Inclusion Criteria – All of the following must be met for tirzepatide to be prescribed:
- Diagnosis of Type II diabetes
- A BMI of 25 or greater
- Inadequate glycemic control on at least 1mg of semaglutide injection plus two or more glucose-lowering drugs
- Change needed to achieve goal A1C is less than 1%.
- Goal A1C should be based on those recommended in the Diabetic Guidelines.
- Adherence to current diabetic medications as evidenced by a review of the prescription refill history during the six months.
Additional Inclusion Criteria – All of the following must be met for tirzepatide to be prescribed:
- Patients with atherosclerotic cardiovascular disease or chronic kidney disease
- Patients of childbearing potential who are using oral contraceptives
Inclusion Criteria for Weight Loss
- BMI of >30 or >27 with patient weight conditions.

Self Quiz
Ask yourself...
- Would a patient with a BMI of 23 with no comorbidities qualify to use tirzepatide to lose 5-10% of their body weight? Why not?
- What impact can tirzepatide have on a person with a healthy weight and BMI of <25?
Considerations for Providers
There are specific considerations that prescribers must be aware of when contemplating if a patient should receive the medication tirzepatide. The following is imperative and must be considered each time the medication is prescribed to a patient:
- Clinical Indications – indicated for treating adults with insufficiently controlled diabetes mellitus as an add-on therapy to diet and exercise; as monotherapy when metformin is considered inappropriate due to contraindications or intolerance; and other medicinal products for treating Type II diabetes.
- Monitoring of medication – routine monitoring of serum calcitonin or thyroid ultrasound is of uncertain value but is recommended for early detection of Medullary Thyroid Cancer (MTC).
- Cost – the average price for tirzepatide ranges from $1,071-$1,351 without any coupons or insurance. Savings Card – manufacturer provided; patients can pay as little as $25 monthly for up to 12 injections. Savings Card – manufacturer provided; patients can pay as little as $25 monthly for up to 12 injections.
- Benefits and Risks – One must evaluate the effectiveness of diabetes and the weight loss experienced. Some of the risks must be evaluated, such as increased cost of medication, unpleasant gastrointestinal side effects, poor insurance coverage, and drug shortages. The FDA has warned that the medicine can cause thyroid C-cell tumors in rats, and it is not sure whether tirzepatide causes similar tumors.
How long does it take for tirzepatide to begin working?
Tirzepatide will start to lower one's blood sugar levels immediately, but it can take 8 to 12 weeks to reach one's target A1C goal.
Compared to other diabetic treatments, studies have shown that it can take eight weeks to reach an A1C target of less than or equal to 7% and 12 weeks to get an A1C of less than or equal to 6.5%. Significant weight loss can occur as early as 28 weeks.
Safe Administration
It is essential to follow the correct steps for safe administration of tirzepatide as listed below:
- The recommended starting dosage is 2.5mg, injected subcutaneously once weekly. The 2.5mg dosage is for treatment initiation and not for glycemic control.
- After four weeks, increase the dosage to 5mg, injected subcutaneously once weekly.
- If additional glycemic control is needed, increase the dosage in 2.5mg increments after at least four weeks on the current dose.
- The maximum dosage is 15mg, injected subcutaneously once weekly.
- If a dose is missed, instruct patients to administer it as soon as possible, within four days (96 hours) after the missed dose. If more than four days have passed, skip the missed dose, and administer the next dose on the regularly scheduled day. In each case, patients can then resume their regular once-weekly dosing schedule.
- The day of weekly Administration can be changed, if necessary, as long as the time between the two doses is at least three days (72 hours).
- Before initiation, train patients and caregivers on proper injection techniques.
- Instruct patients using the single-dose vial to use a syringe appropriate for dose administration (e.g., a 1ml syringe capable of measuring a 0.5 mL dose).
- Administer the medication once weekly, any time of day.
- Inject the medication subcutaneously in the abdomen, thigh, or upper arm.
- Rotate injection sites with each dose.
- Inspect the medication visually before use. It should appear clear and colorless to slightly yellow. Do not use the medicine if particulate matter or discoloration is seen.
- When using the medication with insulin, administer it as separate injections and never mix. It is acceptable to inject tirzepatide and insulin in the same body region, but the injections should not be adjacent.
Does the tirzepatide injection hurt when administered?
Pain from the injection site has not been reported as a common side effect, but it may occur.
Due to the injection being given subcutaneously, slight pain or discomfort can occur.

Self Quiz
Ask yourself...
- The patient asks you," How long will this take to work?" How will you respond?
- The patient reports they have never used an injection before; what methods can you use to teach your patient how to administer this medication safely?
Alternatives to Tirzepatide for Weight Loss Management
In some instances, patients need to be aware of alternatives to tirzepatide in case they cannot take the actual injection for whatever reason. In cases such as these, there are alternative supplements that can be purchased over the counter, and they include the following (7):
- PhenQ – top OTC choice – comprehensive weight loss solution that targets specific body regions, facilitates prompt fat loss, and expedites the weight loss journey.
- PhenGold – the most potent OTC weight loss alternative – one of the top weight loss supplements that boost metabolism, making one less hungry, less tired, and an overall improved feeling.
- Capsiplex BURN – the best choice for men – helps to burn fat faster and keep blood sugar levels in check. It helps to keep one's muscles, curbs hunger, gives one more energy, and torches stubborn fats.
- Trimtone – the best choice for women – helps women to lose weight, eat less, increase metabolism, burn extra calories, and boost energy.
- Prime Shred – best fat burner for men – boosts metabolism, keeps muscles intact, increases energy, and helps maintain focus.
The advanced practicing nurse or prescriber needs to inform patients about alternative options such as these in an effort for individuals to understand that other choices are available and can be used. Many individuals need to be more knowledgeable about alternatives besides tirzepatide due to the extra hype from social media sources that promote advertisements related to tirzepatide only but do not mention the other options.
Why does social media influence and encourage patients to take tirzepatide?
Social media trends can be helpful but can also become harmful by setting unrealistic expectations and promoting a diet culture mentality. They can create an unhealthy obsession with "clean" eating, especially in the younger populations.
Due to this, many individuals take the medication despite any occurrence or history of Type II diabetes, and the drug can ultimately become misused.
It has been noted that there is an influx of patients requesting this medication for weight loss instead of the intended purpose, which is to help control Type II diabetes.
Tirzepatide represents one of the most recent non-medical treatments aimed at managing the symptoms of Type II diabetes. While it is not indicated for weight management, diabetic patients who receive it frequently report a significant reduction in body weight.
Empirical evidence suggests the efficacy of tirzepatide in weight management, and certain physicians currently endorse the Administration of the medication as a therapeutic and effective means to overcome obesity.
What are some severe side effects of tirzepatide that can impact patient safety?
The Administration of tirzepatide can benefit many individuals, but some severe side effects must be mentioned.
These include thyroid tumors, thyroid cancer, pancreatitis, hypoglycemia, serious allergic reactions, kidney issues, severe stomach problems, vision changes, and gallbladder issues. All these side effects must be taken seriously and reported, as they can lead to life-threatening

Self Quiz
Ask yourself...
- With what you have learned in this course, what education will you provide to patients requesting this medication for weight loss?
- Have you seen increased demand for this medication in your current practice?
- If you Google tirzepatide, your results will likely include links to telehealth services promoting this weight-loss medication. To determine eligibility, what special considerations need to be taken to assess a telehealth patient?
Conclusion
Medications like tirzepatide are game changers for those patients with type 2 diabetes that have failed other medications. Unfortunately, several companies seek to profit from its weight-loss benefits through aggressive marketing campaigns that limit the available supply and increase the costs for those who need it. As healthcare providers, we need to use sound clinical judgment and follow the exclusion/inclusion criteria and other guidelines before prescribing this medication, so we do not unintentionally cause harm while looking to appease our patients who request this.
Semaglutide and Type 2 Diabetes
Introduction
In 2017, the FDA approved the semaglutide injectable (Ozempic) for treating type 2 diabetes. The drug has experienced widespread acceptance due to its positive effects on weight loss and lowering of chronic health risks. The drug has risen in popularity over the past few years, as many well-known actors/actresses/songwriters, and more came forward, publicly sharing their weight loss journey.
This rise in popularity has also resulted in significant shortages of this medication, negatively impacting the lives of the diabetic community, local pharmacies, and healthcare providers. The goal of this continuing education course is to educate and empower the healthcare provider in all aspects of this drug regimen: clinical indications, patient education, cost options, and benefit/risk analysis.
Diabetes Overview
Diabetes is a chronic medical condition. Despite advances in diet, medications, and monitoring devices, diabetes diagnoses continue to grow at staggering rates. The Institute for Health Metrics and Evaluation (IHME) reports that over 529 million people worldwide are currently living with diabetes, and that number is expected to grow to 1.3 billion in only 30 years. While the risk factors for diabetes are vast in number (poor diet, inadequate activity, obesity, sedentary lifestyles, daily stressors, and more), the sad reality is that this chronic medical condition will most likely linger on for generations to come despite our efforts to contain this health epidemic (1).
According to the latest research on diabetes, there are over 37 million people in the United States alone with diabetes as of 2022. Statistically, approximately 28 million of them have a confirmed diagnosis, while another estimated 8 million are experiencing symptoms, without an official diagnosis. Diabetes currently ranks as the 7th leading cause of death in the United States (2).

Self Quiz
Ask yourself...
- As a healthcare provider, what has been your experience with treating chronic medical conditions?
- Why do you think there is a continued increase in diabetes, despite advances in medication and monitoring devices to treat this condition?
- Are you currently offering comprehensive care to your patients, including medication, diet, and activity counseling for their chronic health conditions?
Types of Diabetes
In basic terms, diabetes is an impairment in one’s ability to either adequately produce or utilize insulin, which results in elevated levels of circulating glucose. Chronically elevated glucose levels affect blood vessels at every level, causing chronic inflammation and raising the risk of heart disease, stroke, blindness, and atraumatic amputations.
There are three main types of diabetes:
Type 1 diabetes is thought to be an autoimmune disease. Approximately 5-10 percent of people with diabetes are diagnosed with type 1 diabetes. The diagnosis usually occurs in early childhood, and results in a lifetime use of insulin to regulate blood glucose levels.
Type 2 diabetes is thought to be related to dietary and lifestyle choices. It accounts for nearly 90-95 percent of diabetes diagnoses. Usually occurring later in life (adult-elderly population), it is believed to be related to factors such as diet, activity, weight gain, and related factors. Type 2 diabetes is usually controlled by diet and exercise, in addition to oral medications, although injectable insulin may be included in the treatment plan.
Gestational diabetes refers to elevated glucose levels occurring during pregnancy for patients who are not diabetic at the onset of pregnancy. This version of diabetes usually resolves itself post-partum, although a woman may develop type 2 diabetes later in life, unrelated to pregnancy.
Type 2 diabetes in children: no longer a “later in life diagnosis”
Children are now being diagnosed with type 2 diabetes at an alarming rate. Despite widespread education and an increased awareness of diabetes, our up-and-coming generation is unhealthier than ever. Many families lack access to healthy food for their families, due to both general socioeconomic challenges and an increased rate of food insecurity. (19)
The CDC recommends care providers have resources for diabetic patients and their families, such as food and nutrition programs, budget-friendly diabetes meal plans, how to save money on diabetes care, and coping strategies for diabetes. (19)


Self Quiz
Ask yourself...
- Are you able to articulate the different types of diabetes to patients?
- What resources can you offer to the families of children with type 2 diabetes?
Diabetes Signs and Symptoms, Diagnostic Testing
There are various ways to test for diabetes. The fasting blood sugar (FBS)/ fasting glucose level is a simple way to test for diabetes.
The normal fasting glucose level is below 100mg/dl. The fasting glucose result of 100-125mg/dl indicates prediabetes and results above 126mg/dl indicate diabetes.
The hemoglobin A1C blood test is another test used to confirm the diagnosis of diabetes. The patient does not need to be fasting for this test; thus, it is easier to order this test regardless of the time of day. This blood test reflects the average glucose level over the period of 2-3 months.
The normal A1C level is below 5.7%. Test results between 5.7%- 6.4% indicate prediabetes. Test results above 6.5% indicate diabetes.
A random glucose reading above 200mg/dl, done at any time of day, indicates diabetes.
The diagnosis of diabetes is by blood tests, and for improved accuracy, should be based on two separate readings, done (at least) a day apart. In the case of fasting and random blood tests, dietary intake (large amounts of carbohydrates in a single meal) may adversely affect test results. This is not the case when using A1C testing for a confirmation diagnosis, as the results are the average of a 2–3-month span.
Target blood levels for a person with diabetes (3).
Target blood glucose levels for people with diabetes are as follows:
- Fasting glucose 80-130mg/dl.
- Postprandial blood glucose level- less than 180mg/dl
- A1C level 7-8%.
These target ranges are general guidelines. Patient-specific ranges will be dependent on a variety of factors, including preexisting comorbidities, overall health status, age, and activity levels.

The hallmark signs/symptoms of diabetes
- Polyuria- increased urination
- Polydipsia- increased thirst
- Polyphagia-increased hunger/appetite
The truth is, as healthcare providers, you will have patients who have no hallmark signs and symptoms of diabetes; the diagnosis will be found during annual preventive examinations often unrelated to any chronic disease. For this reason, many insurance companies now cover numerous preventive screenings, including diabetes screenings, as part of their wellness and prevention initiatives. These tests are often approved based on a patient's age, or preexisting conditions, rather than outright signs and symptoms.

Self Quiz
Ask yourself...
- What are the typical glucose levels for non-diabetic versus diabetic patients?
- What are the hallmark symptoms you can identify when treating a potentially diabetic patient?
Lifestyle Interventions and the Diabetes Prevention Program
The initial diagnosis of diabetes can be managed in a variety of ways, depending on the severity of the illness at the time of diagnosis. Lifestyle interventions (behavior modification education) are of utmost importance in the care and management of people with diabetes. Research over the past few decades has consistently shown that such interventions have immense positive effects on the successful long-term management of diabetes.
The official Diabetes Prevention Program was created in 2010 (4) and confirmed the effects of lifestyle interventions in the management of diabetes: Lifestyle interventions decreased the incidence of type 2 diabetes by 58% compared with 31% in the metformin-treated group. Thus, these findings now serve as the blueprint, if you will, for all-inclusive, patient-specific disease management guidelines. These lifestyle interventions will be discussed in detail later in the program.
Additional Resources on Diabetes Prevention

Self Quiz
Ask yourself...
- How do lifestyle interventions compare to other kinds of treatment for patients with type 2 diabetes?
Semaglutide
Semaglutide is an injectable drug used in the treatment of type 2 diabetes. It was approved by the FDA in May of 2017.
It is a once-a-week injectable and belongs to the drug class known as glucagon-like peptide-1 receptor agonists (GLP-1RAs) (5). It has been referred to as a “miracle weight loss drug” among those who are living with obesity, despite frequent side effects, unusually high out-of-pocket costs, drug shortages, and weight regain when attempting to stop using the medication.
GLP-1 receptor agonist: Hormone Review
GLP-1 RAs are a class of medications used to treat Type 2 diabetes, and in some cases, obesity treatment. They are also known as GLP-1 receptor agonists, incretin mimetics, and GLP-1 analogs.
Ghrelin and Leptin (6)
Ghrelin and Leptin are two hormones that greatly influence appetite and the sensation of fullness. Often referred to as the “hunger hormone.” Ghrelin is responsible for many functions, including playing a key role in metabolism through glucose and insulin regulation.
Ghrelin, produced in your stomach, signals your brain when you are hungry, and results in increased food intake.
Leptin, conversely, is produced in your fat cells, and signals to the brain when you have eaten enough (by a decrease in appetite).
Glucagon-like peptide-1 receptors
Known as GLP1 receptors, Glucagon-like peptide-1 receptor proteins are located in the beta cells of the pancreas as well as in the neurons in the brain. GLP-1 receptors are involved in the regulation of blood glucose levels and affect the secretion of insulin. These cells encourage the release of insulin from the pancreas, increase the volume of beta cells, and reduce the release of glucagon. In doing so, they increase the feeling of fullness during and between meals, suppressing the appetite and slowing gastric emptying.

Self Quiz
Ask yourself...
- What are some problems patients might face if they choose to take semaglutide?
- How do Ghrelin and Leptin relate to a patient's appetite?
What is meant by receptor agonist and antagonist?
The term agonist refers to any substance that mimics the actions of a hormone in producing a specific response: a receptor antagonist blocks a response from occurring.
Opioids are examples of receptor agonists in that they produce responses such as analgesia.
Naloxone/Narcan is an example of a receptor antagonist, in that it binds to a receptor site and decreases/blocks a response from occurring.
Semaglutide mechanism of action (7)
GLP-1 agonists work in several ways to positively affect glucose levels. Their mechanism of action includes the following:
- Increasing (stimulating) insulin secretion by the pancreatic beta cells.
- Decreasing the production of glucagon, a hormone that raises blood glucose levels
- Decreasing (slowing) gastric emptying
- Decreasing appetite (and thereby reducing food intake) by creating a sensation of stomach fullness
Through these mechanisms of action, semaglutide results in a lowering of serum glucose/A1C levels, which lowers the risk of cardiovascular events. Studies have also shown that semaglutide resulted in weight loss (approximately 8-14 pounds on average {dose dependent results}.

Self Quiz
Ask yourself...
- What is the difference between an agonist and antagonist substance?
- How much weight do patients lose, on average, when taking semaglutide?
Side Effects of Semaglutide
Common side effects of semaglutide (8)
Common side effects may include any of the following:
- Nausea and vomiting
- Headache
- Diarrhea and stomach pain
- Upset stomach, indigestion, constipation, flatulence
These side effects usually subside within a few weeks, as the patient becomes acclimated to the medication.
Serious side effects of semaglutide
- Hypoglycemia- enhanced/worsened when used in combination with other diabetes medication. Symptoms may include drowsiness, confusion, weakness, irritability, and headache.
- Symptoms may include abdominal pain and distension, nausea and vomiting, fever, and back pain.
- Diabetic retinopathy. Symptoms may include blurred vision, vision loss, and diminished night vision.
- Kidney damage/injury/failure. Symptoms may include fatigue, nausea, diminished urine output, confusion, and edema of extremities.
- Gallbladder disease. Symptoms may include gallstones, abdominal pain, nausea and vomiting, and poor appetite.
Black Box Warning (9)
Semaglutide has a Black Box Warning for thyroid cancer. This is the most serious warning from the Food and Drug Administration (FDA) and is intended to alert consumers to the potential risks of a medication. This black box warning was issued when research found that the drug increased the risk of thyroid tumors in animals.
It is not known if semaglutide actually causes tumors in humans.
Contraindications
- Semaglutide is contraindicated in people with a personal or family history of MTC (medullary thyroid cancer) or in patients with multiple endocrine neoplasia syndrome type 2.
- Known hypersensitivity to semaglutide or any of the product components
Cautions
As noted under “serious side effects”, there have been reports of new illnesses or worsening of existing health conditions occurring “post-marketing”. Thus, healthcare providers are strongly encouraged to continue ongoing surveillance of any patients on semaglutide therapy. In addition, there is insufficient data available regarding the use of semaglutide by pregnant women. Women are therefore highly encouraged to stop any treatment with semaglutide for at least 2 months prior to a planned pregnancy.

Self Quiz
Ask yourself...
- Can you name the 4 common side effects of semaglutide?
- What is the most severe warning associated with semaglutide?
Dosing
Semaglutide is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus (T2DM). It is looked upon favorably to reduce the risk of cardiovascular events in adults with T2DM and a preexisting history of cardiovascular disease. This drug is FDA-approved for use in people with diabetes, with a BMI of 27% or higher (a BMI of 25-29.9% is considered overweight).
Semaglutide (Ozempic) is available as an injectable prescription medication. Doses include 0.5mg, 1mg, or 2 mg, once weekly.
The injection should be administered subcutaneously to the abdomen, thigh, or upper arm. Injection sites should be rotated, and given as a single injection.
Start at 0.25 mg once weekly. After 4 weeks, increase the dose to 0.5 mg once weekly.
- If additional glycemic control is needed, increase the dose to 1 mg once weekly after at least 4 weeks on the 0.5 mg dose.
- If additional glycemic control is needed, increase the dose to 2 mg once weekly after at least 4 weeks on the 1 mg dose
Administer once weekly at any time of day, with or without meals. The maximum dose recommendation is 2mg/weekly once weekly.
Note: The initial 0.25-mg dose is intended for treatment initiation and is not effective for glycemic control
Missing Dose Guidelines
- If the missed dose is ≤5 days: Administer dose as soon as possible
- If missed dose >5 days: Skip the missed dose and administer the next dose on the regularly scheduled day; patients can then resume their regular once-weekly dosing schedule
Administration Day Guidelines (10).
The administration day each week can be changed, if necessary, as long as the time between 2 doses is at least 2 days (>48 hours)
Dose Availability (packaging)
- 2mg/1.5mL (1.34mg/mL); delivers doses of 0.25mg or 0.5mg per injection or four to eight doses per injection pen
- 4mg/3mL (1.34mg/mL); delivers 1mg per injection or 4 doses per injection pen
- 8mg/3mL (2.68 mg/mL); delivers 2mg per injection or 4 doses per injection pen
Treatment Goals- Effects on A1C and Weight (11)
A majority of adults who were placed on injectable semaglutide for diabetes management achieved a target A1C under 7% and were able to maintain it.
- Dose specific effects on A1C were as follows:
- 0.5mg dose injection yielded a 1.4% decrease
- 1.0mg dose injection yielded a 1.6% decrease
- 2.0 mg dose injection, in combination with diabetes pills, yielded a 2.1% decrease in A1C.
Adults taking semifluid injectables for diabetes management also noted weight loss.
- 8-pound weight loss reported with 0.5mg dose injection
- 10 pounds weight loss reported with 1.0mg dose injection
- Up to 14 pounds of weight loss reported with a 2.0mg dose injection


Self Quiz
Ask yourself...
- What should you tell a patient if they miss their injection by more than 5 days? What if it has been less than five days?
Prescribing insights: Long-Term therapy for a chronic condition?
Semaglutide is viewed favorably as a treatment option for Type 2 diabetes. It appears to lower A1C levels and body weight in the majority of patients, lowering their risk of future cardiovascular events.
The question of long-term medication use, for a chronic health condition, is being heavily discussed in the media. While a percentage of people can decrease or eliminate the need for chronic medications through significant lifestyle changes, there have been reports of weight gain in those who stopped taking this injectable medication.
Without intense lifestyle behavior modification education, there is a heightened risk of weight regain in the absence of such medications. Leaders in the treatment of obesity and related illnesses have commented that this drug is intended for long-term use.
Examples of this include the following:
“GLP-1 medications [like Ozempic] are designed to be taken long-term... They are chronic medications for the treatment of chronic conditions (both diabetes and obesity) (12)". - Christopher McGowan, M.D., a gastroenterologist specializing in obesity medicine and endobariatrics
“As with many chronic conditions, most people who use the drugs for diabetes or weight loss will need to keep taking them to keep benefiting from them. Depending on your individual situation, and without sustained lifestyle changes, it is likely you would need to be on these medications indefinitely to maintain weight loss (13)." - Dr. Cecilia Low Wang, a UCHealth expert in endocrinology, diabetes and metabolism.

Self Quiz
Ask yourself...
- Is semaglutide considered to be a long-term treatment for type 2 diabetes?
Cost Concerns
At this time, injectable semaglutide, FDA-approved for the treatment of Type 2 diabetes, has a self-pay price tag of $935.77 per month (4 injections). With FDA approval, many people with diabetes, insured under commercial plans, receive the drug for the cost of their copay. Those patients without coverage may use pharmacy discount cards that reduce the price, on average, to $814.55/month.
The following links are available to familiarize yourself with patient assistance programs related to semaglutide injectables.
Semaglutide Cost Savings Programs
The following links are provided to explore various semaglutide cost savings programs.

Self Quiz
Ask yourself...
- What resources can you offer patients who are struggling to pay for semaglutide?
Emerging Concerns: Semaglutide and gastroparesis
In August 2023, a first-of-its-kind lawsuit was filed in Louisiana, against the makers of semaglutide. The lawsuit states the makers of this injectable drug did not adequately warn patients about the risk of severe gastrointestinal issues/possible gastroparesis.
The plaintiff in this case had used both Ozempic and Mounjaro and experienced repeated episodes of severe gastrointestinal events, warranting trips to the emergency room and additional medications to alleviate her symptoms (14). While this lawsuit is in the developing stages, it bears mentioning in terms of concerns over long-term usage of the drug and possible complications.
While the drug labeling for semaglutide (Ozempic) does not specifically mention gastroparesis, the semaglutide/Mounjaro drug label does state that the drug has not been studied in patients with severe gastrointestinal disease and is therefore not recommended in these patients.
Up to 50% of people with diabetes have some degree of delayed gastric emptying, but most have no digestive symptoms or have only mild symptoms. For some people with diabetes, problems managing blood glucose levels may be a sign of delayed gastric emptying (15).
Healthcare providers should evaluate all patients with diabetes for possible symptoms of underlying gastroparesis, such as the feeling of fullness shortly after beginning a meal, or the inability to finish a regular meal. Other symptoms of gastroparesis may include abdominal pain, nausea, bloating, vomiting, and anorexia.
Diabetes and gastroparesis
Uncontrolled or poorly controlled diabetes can affect nerve endings systemwide. Diabetes is a very common cause of gastroparesis. Although the condition is rare it occurs more often in people with chronic conditions such as diabetes, autoimmune diseases, and nervous system disorders. Nerve endings are injured or damaged, cease to function properly, and result in delayed gastric emptying. The delay in gastric emptying can cause various symptoms, such as nausea, vomiting, bloating and distension, abdominal pain, and poor appetite.
In addition to underlying medical conditions, some medications may cause symptoms of gastroparesis (delays in gastric emptying and overall gastric motility. These medications include narcotics, antidepressants, and anticholinergics.
Left untreated, diabetic gastroparesis may lead to malnutrition, electrolyte imbalances, and poor glucose management and control.

Self Quiz
Ask yourself...
- Why should nurses prescribing semaglutide watch out for symptoms of gastroparesis?
- What do you think are some ethical issues with semaglutide use for weight loss?
Diabetes Lifestyle changes: Patient education (16)
- Weight Management
- Healthy Eating
- Physical Activity
- Smoking Cessation
- Stress Management
The importance of patient education regarding lifestyle changes is a priority. As with any chronic medication condition, the patient and their family/support system must be given every opportunity to educate and empower themselves on self-management of their disease process. Patients must be given the benefit of the doubt that they can indeed embrace their health and well-being and work with their healthcare provider in maximizing their health outcomes.
For diabetes mellitus, numerous lifestyle behaviors should be addressed and actively worked on, so that the patient receives the maximum health benefits. The following lifestyle behaviors are in no particular order; they all warrant discussion at every office visit.
Diet
A person with diabetes should be educated on the effects of food and nutrition on their glucose level. Referrals to a dietitian/nutritionist or Certified Diabetes Care Education Specialist (CDCES) should be considered a top priority. Well-balanced nutritional intake, appropriate carbohydrate awareness, calorie monitoring if weight loss is appropriate to your specific patient) and medication/food interactions are all essential aspects of dietary lifestyle education. Many commercial insurance plans, as well as hospital community outreach programs, offer diabetes self-management classes.

Activity (17)
The CDC recommends a target goal of 150 minutes weekly, Patients should be educated on the positive effects of daily activity on overall health and well-being, stress management, and metabolism. Patients should find activities they are genuinely interested in, involve family and friends, and slowly build greater endurance through increased intervals of longer duration.
Sleep hygiene (18)
Patients should be educated on the positive effects of a good night’s sleep. The aim should be approximately 7-8 hours of restful sleep. Electronics should be powered down and (optimally) removed from the bedroom. A dark, well-vented, cool room temperature is encouraged, and large meals and late-evening caffeine should be avoided.
Medication adherence/ literacy
Medication education is critical to the health and well-being of a patient. Routine education of the patient, and family members or support systems when available, should be supportive and patient-specific. Patients should be assessed on language barriers, literacy issues, and related comprehension concerns. Medication education should include effects, side effects, treatment goals, and sick day management. Emergency care issues should also be discussed. Any monitoring equipment (continuous glucose monitors, accuchecks, lancets) should be reviewed with patients and confirmed with return verbalization and demonstration.
As discussed in this course, patients with chronic diseases must learn self-management techniques to optimize their health and well-being. They must become confident in their understanding of their disease process and take ownership of their health. In doing so, they minimize the risk of long-term complications, improve their self-worth, and actively invest (both time and money) in their future.

Self Quiz
Ask yourself...
- How does sleep, diet, and activity level affect the treatment of type 2 diabetes?
Ozempic Case Study
- 52-year-old female
- Height 67 inches
- Weight 225 pounds
- B/P 138/84, Heart rate 76 NSR
- BMI 35.2%
- Nonsmoker, occasional social drinker
- Multiple attempts at dieting without success.
- Diagnosed T2DM approx. 6 months ago current A1C 7.5%; initial medication Metformin 500mg BID tablets; tolerated well. No GI upsets.
Today’s appointment is for evaluation and additional medication consideration (the patient requested this appointment)
The patient was diagnosed with T2DM approximately 6 months ago. Initial A1C 8.0%. Current A1C 7.7%
Despite an improved diet and adherence to the medication regimen, the patient voiced frustration at the lack of weight loss. Requesting additional medication. Has a neighbor friend who began injectable Ozempic and is having “really great results with it. I want to start on it as well”.
- What are your thoughts on prescribing semaglutide injectable for this patient?
- What objective health data points should be taken into consideration regarding prescribing semaglutide for this patient?
The patient has expressed frustration that despite taking her medications and adjusting her diet, she has not lost any weight in the past 6 months. She has “heard from her neighbor friend that the weight just melts off immediately” and she is ready to start this medication.
- What concerns do you know about this patient's understanding of weight loss as it relates to semaglutide?
- What prescribing information, specific to semaglutide and weight loss, could you share with your patient regarding realistic weight loss targets?
- In addition to teaching your patient proper injection technique for the use of semaglutide, what other lifestyle education behaviors should you discuss at this point?
- What information should you share with your patient regarding the long-term use of semaglutide and the potential risks of stopping this medication (as it relates to weight regain)?
Your patient decides to go ahead with the semaglutide regimen.
- What are some patient education guidelines regarding common side effects of this medication?
- How often is the dose increased? What is the maximum dose this patient can receive weekly?
Your patient wants to know how long she will be taking this medication.
- What talking points will you cover regarding the long-term use of this medication?
- How do you best prepare this patient for long-term success with this medication?
- What lifestyle behavior modification education would you discuss with your patient, to give her the best chance at successfully managing her diabetes?
Medication Assisted Treatment
Introduction
In 2017, the FDA approved the semaglutide injectable (Ozempic) for treating type 2 diabetes. The drug has experienced widespread acceptance due to its positive effects on weight loss and lowering of chronic health risks. The drug has risen in popularity over the past few years, as many well-known actors/actresses/songwriters, and more came forward, publicly sharing their weight loss journey.
This rise in popularity has also resulted in significant shortages of this medication, negatively impacting the lives of the diabetic community, local pharmacies, and healthcare providers. The goal of this continuing education course is to educate and empower the healthcare provider in all aspects of this drug regimen: clinical indications, patient education, cost options, and benefit/risk analysis.
Diabetes Overview
Diabetes is a chronic medical condition. Despite advances in diet, medications, and monitoring devices, diabetes diagnoses continue to grow at staggering rates. The Institute for Health Metrics and Evaluation (IHME) reports that over 529 million people worldwide are currently living with diabetes, and that number is expected to grow to 1.3 billion in only 30 years. While the risk factors for diabetes are vast in number (poor diet, inadequate activity, obesity, sedentary lifestyles, daily stressors, and more), the sad reality is that this chronic medical condition will most likely linger on for generations to come despite our efforts to contain this health epidemic (1).
According to the latest research on diabetes, there are over 37 million people in the United States alone with diabetes as of 2022. Statistically, approximately 28 million of them have a confirmed diagnosis, while another estimated 8 million are experiencing symptoms, without an official diagnosis. Diabetes currently ranks as the 7th leading cause of death in the United States (2).

Self Quiz
Ask yourself...
- As a healthcare provider, what has been your experience with treating chronic medical conditions?
- Why do you think there is a continued increase in diabetes, despite advances in medication and monitoring devices to treat this condition?
- Are you currently offering comprehensive care to your patients, including medication, diet, and activity counseling for their chronic health conditions?
Types of Diabetes
In basic terms, diabetes is an impairment in one’s ability to either adequately produce or utilize insulin, which results in elevated levels of circulating glucose. Chronically elevated glucose levels affect blood vessels at every level, causing chronic inflammation and raising the risk of heart disease, stroke, blindness, and atraumatic amputations.
There are three main types of diabetes:
Type 1 diabetes is thought to be an autoimmune disease. Approximately 5-10 percent of people with diabetes are diagnosed with type 1 diabetes. The diagnosis usually occurs in early childhood, and results in a lifetime use of insulin to regulate blood glucose levels.
Type 2 diabetes is thought to be related to dietary and lifestyle choices. It accounts for nearly 90-95 percent of diabetes diagnoses. Usually occurring later in life (adult-elderly population), it is believed to be related to factors such as diet, activity, weight gain, and related factors. Type 2 diabetes is usually controlled by diet and exercise, in addition to oral medications, although injectable insulin may be included in the treatment plan.
Gestational diabetes refers to elevated glucose levels occurring during pregnancy for patients who are not diabetic at the onset of pregnancy. This version of diabetes usually resolves itself post-partum, although a woman may develop type 2 diabetes later in life, unrelated to pregnancy.
Type 2 diabetes in children: no longer a “later in life diagnosis”
Children are now being diagnosed with type 2 diabetes at an alarming rate. Despite widespread education and an increased awareness of diabetes, our up-and-coming generation is unhealthier than ever. Many families lack access to healthy food for their families, due to both general socioeconomic challenges and an increased rate of food insecurity. (19)
The CDC recommends care providers have resources for diabetic patients and their families, such as food and nutrition programs, budget-friendly diabetes meal plans, how to save money on diabetes care, and coping strategies for diabetes. (19)


Self Quiz
Ask yourself...
- Are you able to articulate the different types of diabetes to patients?
- What resources can you offer to the families of children with type 2 diabetes?
Diabetes Signs and Symptoms, Diagnostic Testing
There are various ways to test for diabetes. The fasting blood sugar (FBS)/ fasting glucose level is a simple way to test for diabetes.
The normal fasting glucose level is below 100mg/dl. The fasting glucose result of 100-125mg/dl indicates prediabetes and results above 126mg/dl indicate diabetes.
The hemoglobin A1C blood test is another test used to confirm the diagnosis of diabetes. The patient does not need to be fasting for this test; thus, it is easier to order this test regardless of the time of day. This blood test reflects the average glucose level over the period of 2-3 months.
The normal A1C level is below 5.7%. Test results between 5.7%- 6.4% indicate prediabetes. Test results above 6.5% indicate diabetes.
A random glucose reading above 200mg/dl, done at any time of day, indicates diabetes.
The diagnosis of diabetes is by blood tests, and for improved accuracy, should be based on two separate readings, done (at least) a day apart. In the case of fasting and random blood tests, dietary intake (large amounts of carbohydrates in a single meal) may adversely affect test results. This is not the case when using A1C testing for a confirmation diagnosis, as the results are the average of a 2–3-month span.
Target blood levels for a person with diabetes (3).
Target blood glucose levels for people with diabetes are as follows:
- Fasting glucose 80-130mg/dl.
- Postprandial blood glucose level- less than 180mg/dl
- A1C level 7-8%.
These target ranges are general guidelines. Patient-specific ranges will be dependent on a variety of factors, including preexisting comorbidities, overall health status, age, and activity levels.

The hallmark signs/symptoms of diabetes
- Polyuria- increased urination
- Polydipsia- increased thirst
- Polyphagia-increased hunger/appetite
The truth is, as healthcare providers, you will have patients who have no hallmark signs and symptoms of diabetes; the diagnosis will be found during annual preventive examinations often unrelated to any chronic disease. For this reason, many insurance companies now cover numerous preventive screenings, including diabetes screenings, as part of their wellness and prevention initiatives. These tests are often approved based on a patient's age, or preexisting conditions, rather than outright signs and symptoms.

Self Quiz
Ask yourself...
- What are the typical glucose levels for non-diabetic versus diabetic patients?
- What are the hallmark symptoms you can identify when treating a potentially diabetic patient?
Lifestyle Interventions and the Diabetes Prevention Program
The initial diagnosis of diabetes can be managed in a variety of ways, depending on the severity of the illness at the time of diagnosis. Lifestyle interventions (behavior modification education) are of utmost importance in the care and management of people with diabetes. Research over the past few decades has consistently shown that such interventions have immense positive effects on the successful long-term management of diabetes.
The official Diabetes Prevention Program was created in 2010 (4) and confirmed the effects of lifestyle interventions in the management of diabetes: Lifestyle interventions decreased the incidence of type 2 diabetes by 58% compared with 31% in the metformin-treated group. Thus, these findings now serve as the blueprint, if you will, for all-inclusive, patient-specific disease management guidelines. These lifestyle interventions will be discussed in detail later in the program.
Additional Resources on Diabetes Prevention

Self Quiz
Ask yourself...
- How do lifestyle interventions compare to other kinds of treatment for patients with type 2 diabetes?
Semaglutide
Semaglutide is an injectable drug used in the treatment of type 2 diabetes. It was approved by the FDA in May of 2017.
It is a once-a-week injectable and belongs to the drug class known as glucagon-like peptide-1 receptor agonists (GLP-1RAs) (5). It has been referred to as a “miracle weight loss drug” among those who are living with obesity, despite frequent side effects, unusually high out-of-pocket costs, drug shortages, and weight regain when attempting to stop using the medication.
GLP-1 receptor agonist: Hormone Review
GLP-1 RAs are a class of medications used to treat Type 2 diabetes, and in some cases, obesity treatment. They are also known as GLP-1 receptor agonists, incretin mimetics, and GLP-1 analogs.
Ghrelin and Leptin (6)
Ghrelin and Leptin are two hormones that greatly influence appetite and the sensation of fullness. Often referred to as the “hunger hormone.” Ghrelin is responsible for many functions, including playing a key role in metabolism through glucose and insulin regulation.
Ghrelin, produced in your stomach, signals your brain when you are hungry, and results in increased food intake.
Leptin, conversely, is produced in your fat cells, and signals to the brain when you have eaten enough (by a decrease in appetite).
Glucagon-like peptide-1 receptors
Known as GLP1 receptors, Glucagon-like peptide-1 receptor proteins are located in the beta cells of the pancreas as well as in the neurons in the brain. GLP-1 receptors are involved in the regulation of blood glucose levels and affect the secretion of insulin. These cells encourage the release of insulin from the pancreas, increase the volume of beta cells, and reduce the release of glucagon. In doing so, they increase the feeling of fullness during and between meals, suppressing the appetite and slowing gastric emptying.

Self Quiz
Ask yourself...
- What are some problems patients might face if they choose to take semaglutide?
- How do Ghrelin and Leptin relate to a patient's appetite?
What is meant by receptor agonist and antagonist?
The term agonist refers to any substance that mimics the actions of a hormone in producing a specific response: a receptor antagonist blocks a response from occurring.
Opioids are examples of receptor agonists in that they produce responses such as analgesia.
Naloxone/Narcan is an example of a receptor antagonist, in that it binds to a receptor site and decreases/blocks a response from occurring.
Semaglutide mechanism of action (7)
GLP-1 agonists work in several ways to positively affect glucose levels. Their mechanism of action includes the following:
- Increasing (stimulating) insulin secretion by the pancreatic beta cells.
- Decreasing the production of glucagon, a hormone that raises blood glucose levels
- Decreasing (slowing) gastric emptying
- Decreasing appetite (and thereby reducing food intake) by creating a sensation of stomach fullness
Through these mechanisms of action, semaglutide results in a lowering of serum glucose/A1C levels, which lowers the risk of cardiovascular events. Studies have also shown that semaglutide resulted in weight loss (approximately 8-14 pounds on average {dose dependent results}.

Self Quiz
Ask yourself...
- What is the difference between an agonist and antagonist substance?
- How much weight do patients lose, on average, when taking semaglutide?
Side Effects of Semaglutide
Common side effects of semaglutide (8)
Common side effects may include any of the following:
- Nausea and vomiting
- Headache
- Diarrhea and stomach pain
- Upset stomach, indigestion, constipation, flatulence
These side effects usually subside within a few weeks, as the patient becomes acclimated to the medication.
Serious side effects of semaglutide
- Hypoglycemia- enhanced/worsened when used in combination with other diabetes medication. Symptoms may include drowsiness, confusion, weakness, irritability, and headache.
- Symptoms may include abdominal pain and distension, nausea and vomiting, fever, and back pain.
- Diabetic retinopathy. Symptoms may include blurred vision, vision loss, and diminished night vision.
- Kidney damage/injury/failure. Symptoms may include fatigue, nausea, diminished urine output, confusion, and edema of extremities.
- Gallbladder disease. Symptoms may include gallstones, abdominal pain, nausea and vomiting, and poor appetite.
Black Box Warning (9)
Semaglutide has a Black Box Warning for thyroid cancer. This is the most serious warning from the Food and Drug Administration (FDA) and is intended to alert consumers to the potential risks of a medication. This black box warning was issued when research found that the drug increased the risk of thyroid tumors in animals.
It is not known if semaglutide actually causes tumors in humans.
Contraindications
- Semaglutide is contraindicated in people with a personal or family history of MTC (medullary thyroid cancer) or in patients with multiple endocrine neoplasia syndrome type 2.
- Known hypersensitivity to semaglutide or any of the product components
Cautions
As noted under “serious side effects”, there have been reports of new illnesses or worsening of existing health conditions occurring “post-marketing”. Thus, healthcare providers are strongly encouraged to continue ongoing surveillance of any patients on semaglutide therapy. In addition, there is insufficient data available regarding the use of semaglutide by pregnant women. Women are therefore highly encouraged to stop any treatment with semaglutide for at least 2 months prior to a planned pregnancy.

Self Quiz
Ask yourself...
- Can you name the 4 common side effects of semaglutide?
- What is the most severe warning associated with semaglutide?
Dosing
Semaglutide is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus (T2DM). It is looked upon favorably to reduce the risk of cardiovascular events in adults with T2DM and a preexisting history of cardiovascular disease. This drug is FDA-approved for use in people with diabetes, with a BMI of 27% or higher (a BMI of 25-29.9% is considered overweight).
Semaglutide (Ozempic) is available as an injectable prescription medication. Doses include 0.5mg, 1mg, or 2 mg, once weekly.
The injection should be administered subcutaneously to the abdomen, thigh, or upper arm. Injection sites should be rotated, and given as a single injection.
Start at 0.25 mg once weekly. After 4 weeks, increase the dose to 0.5 mg once weekly.
- If additional glycemic control is needed, increase the dose to 1 mg once weekly after at least 4 weeks on the 0.5 mg dose.
- If additional glycemic control is needed, increase the dose to 2 mg once weekly after at least 4 weeks on the 1 mg dose
Administer once weekly at any time of day, with or without meals. The maximum dose recommendation is 2mg/weekly once weekly.
Note: The initial 0.25-mg dose is intended for treatment initiation and is not effective for glycemic control
Missing Dose Guidelines
- If the missed dose is ≤5 days: Administer dose as soon as possible
- If missed dose >5 days: Skip the missed dose and administer the next dose on the regularly scheduled day; patients can then resume their regular once-weekly dosing schedule
Administration Day Guidelines (10).
The administration day each week can be changed, if necessary, as long as the time between 2 doses is at least 2 days (>48 hours)
Dose Availability (packaging)
- 2mg/1.5mL (1.34mg/mL); delivers doses of 0.25mg or 0.5mg per injection or four to eight doses per injection pen
- 4mg/3mL (1.34mg/mL); delivers 1mg per injection or 4 doses per injection pen
- 8mg/3mL (2.68 mg/mL); delivers 2mg per injection or 4 doses per injection pen
Treatment Goals- Effects on A1C and Weight (11)
A majority of adults who were placed on injectable semaglutide for diabetes management achieved a target A1C under 7% and were able to maintain it.
- Dose specific effects on A1C were as follows:
- 0.5mg dose injection yielded a 1.4% decrease
- 1.0mg dose injection yielded a 1.6% decrease
- 2.0 mg dose injection, in combination with diabetes pills, yielded a 2.1% decrease in A1C.
Adults taking semifluid injectables for diabetes management also noted weight loss.
- 8-pound weight loss reported with 0.5mg dose injection
- 10 pounds weight loss reported with 1.0mg dose injection
- Up to 14 pounds of weight loss reported with a 2.0mg dose injection


Self Quiz
Ask yourself...
- What should you tell a patient if they miss their injection by more than 5 days? What if it has been less than five days?
Prescribing insights: Long-Term therapy for a chronic condition?
Semaglutide is viewed favorably as a treatment option for Type 2 diabetes. It appears to lower A1C levels and body weight in the majority of patients, lowering their risk of future cardiovascular events.
The question of long-term medication use, for a chronic health condition, is being heavily discussed in the media. While a percentage of people can decrease or eliminate the need for chronic medications through significant lifestyle changes, there have been reports of weight gain in those who stopped taking this injectable medication.
Without intense lifestyle behavior modification education, there is a heightened risk of weight regain in the absence of such medications. Leaders in the treatment of obesity and related illnesses have commented that this drug is intended for long-term use.
Examples of this include the following:
“GLP-1 medications [like Ozempic] are designed to be taken long-term... They are chronic medications for the treatment of chronic conditions (both diabetes and obesity) (12)". - Christopher McGowan, M.D., a gastroenterologist specializing in obesity medicine and endobariatrics
“As with many chronic conditions, most people who use the drugs for diabetes or weight loss will need to keep taking them to keep benefiting from them. Depending on your individual situation, and without sustained lifestyle changes, it is likely you would need to be on these medications indefinitely to maintain weight loss (13)." - Dr. Cecilia Low Wang, a UCHealth expert in endocrinology, diabetes and metabolism.

Self Quiz
Ask yourself...
- Is semaglutide considered to be a long-term treatment for type 2 diabetes?
Cost Concerns
At this time, injectable semaglutide, FDA-approved for the treatment of Type 2 diabetes, has a self-pay price tag of $935.77 per month (4 injections). With FDA approval, many people with diabetes, insured under commercial plans, receive the drug for the cost of their copay. Those patients without coverage may use pharmacy discount cards that reduce the price, on average, to $814.55/month.
The following links are available to familiarize yourself with patient assistance programs related to semaglutide injectables.
Semaglutide Cost Savings Programs
The following links are provided to explore various semaglutide cost savings programs.

Self Quiz
Ask yourself...
- What resources can you offer patients who are struggling to pay for semaglutide?
Emerging Concerns: Semaglutide and gastroparesis
In August 2023, a first-of-its-kind lawsuit was filed in Louisiana, against the makers of semaglutide. The lawsuit states the makers of this injectable drug did not adequately warn patients about the risk of severe gastrointestinal issues/possible gastroparesis.
The plaintiff in this case had used both Ozempic and Mounjaro and experienced repeated episodes of severe gastrointestinal events, warranting trips to the emergency room and additional medications to alleviate her symptoms (14). While this lawsuit is in the developing stages, it bears mentioning in terms of concerns over long-term usage of the drug and possible complications.
While the drug labeling for semaglutide (Ozempic) does not specifically mention gastroparesis, the semaglutide/Mounjaro drug label does state that the drug has not been studied in patients with severe gastrointestinal disease and is therefore not recommended in these patients.
Up to 50% of people with diabetes have some degree of delayed gastric emptying, but most have no digestive symptoms or have only mild symptoms. For some people with diabetes, problems managing blood glucose levels may be a sign of delayed gastric emptying (15).
Healthcare providers should evaluate all patients with diabetes for possible symptoms of underlying gastroparesis, such as the feeling of fullness shortly after beginning a meal, or the inability to finish a regular meal. Other symptoms of gastroparesis may include abdominal pain, nausea, bloating, vomiting, and anorexia.
Diabetes and gastroparesis
Uncontrolled or poorly controlled diabetes can affect nerve endings systemwide. Diabetes is a very common cause of gastroparesis. Although the condition is rare it occurs more often in people with chronic conditions such as diabetes, autoimmune diseases, and nervous system disorders. Nerve endings are injured or damaged, cease to function properly, and result in delayed gastric emptying. The delay in gastric emptying can cause various symptoms, such as nausea, vomiting, bloating and distension, abdominal pain, and poor appetite.
In addition to underlying medical conditions, some medications may cause symptoms of gastroparesis (delays in gastric emptying and overall gastric motility. These medications include narcotics, antidepressants, and anticholinergics.
Left untreated, diabetic gastroparesis may lead to malnutrition, electrolyte imbalances, and poor glucose management and control.

Self Quiz
Ask yourself...
- Why should nurses prescribing semaglutide watch out for symptoms of gastroparesis?
- What do you think are some ethical issues with semaglutide use for weight loss?
Diabetes Lifestyle changes: Patient education (16)
- Weight Management
- Healthy Eating
- Physical Activity
- Smoking Cessation
- Stress Management
The importance of patient education regarding lifestyle changes is a priority. As with any chronic medication condition, the patient and their family/support system must be given every opportunity to educate and empower themselves on self-management of their disease process. Patients must be given the benefit of the doubt that they can indeed embrace their health and well-being and work with their healthcare provider in maximizing their health outcomes.
For diabetes mellitus, numerous lifestyle behaviors should be addressed and actively worked on, so that the patient receives the maximum health benefits. The following lifestyle behaviors are in no particular order; they all warrant discussion at every office visit.
Diet
A person with diabetes should be educated on the effects of food and nutrition on their glucose level. Referrals to a dietitian/nutritionist or Certified Diabetes Care Education Specialist (CDCES) should be considered a top priority. Well-balanced nutritional intake, appropriate carbohydrate awareness, calorie monitoring if weight loss is appropriate to your specific patient) and medication/food interactions are all essential aspects of dietary lifestyle education. Many commercial insurance plans, as well as hospital community outreach programs, offer diabetes self-management classes.

Activity (17)
The CDC recommends a target goal of 150 minutes weekly, Patients should be educated on the positive effects of daily activity on overall health and well-being, stress management, and metabolism. Patients should find activities they are genuinely interested in, involve family and friends, and slowly build greater endurance through increased intervals of longer duration.
Sleep hygiene (18)
Patients should be educated on the positive effects of a good night’s sleep. The aim should be approximately 7-8 hours of restful sleep. Electronics should be powered down and (optimally) removed from the bedroom. A dark, well-vented, cool room temperature is encouraged, and large meals and late-evening caffeine should be avoided.
Medication adherence/ literacy
Medication education is critical to the health and well-being of a patient. Routine education of the patient, and family members or support systems when available, should be supportive and patient-specific. Patients should be assessed on language barriers, literacy issues, and related comprehension concerns. Medication education should include effects, side effects, treatment goals, and sick day management. Emergency care issues should also be discussed. Any monitoring equipment (continuous glucose monitors, accuchecks, lancets) should be reviewed with patients and confirmed with return verbalization and demonstration.
As discussed in this course, patients with chronic diseases must learn self-management techniques to optimize their health and well-being. They must become confident in their understanding of their disease process and take ownership of their health. In doing so, they minimize the risk of long-term complications, improve their self-worth, and actively invest (both time and money) in their future.

Self Quiz
Ask yourself...
- How does sleep, diet, and activity level affect the treatment of type 2 diabetes?
Ozempic Case Study
- 52-year-old female
- Height 67 inches
- Weight 225 pounds
- B/P 138/84, Heart rate 76 NSR
- BMI 35.2%
- Nonsmoker, occasional social drinker
- Multiple attempts at dieting without success.
- Diagnosed T2DM approx. 6 months ago current A1C 7.5%; initial medication Metformin 500mg BID tablets; tolerated well. No GI upsets.
Today’s appointment is for evaluation and additional medication consideration (the patient requested this appointment)
The patient was diagnosed with T2DM approximately 6 months ago. Initial A1C 8.0%. Current A1C 7.7%
Despite an improved diet and adherence to the medication regimen, the patient voiced frustration at the lack of weight loss. Requesting additional medication. Has a neighbor friend who began injectable Ozempic and is having “really great results with it. I want to start on it as well”.
- What are your thoughts on prescribing semaglutide injectable for this patient?
- What objective health data points should be taken into consideration regarding prescribing semaglutide for this patient?
The patient has expressed frustration that despite taking her medications and adjusting her diet, she has not lost any weight in the past 6 months. She has “heard from her neighbor friend that the weight just melts off immediately” and she is ready to start this medication.
- What concerns do you know about this patient's understanding of weight loss as it relates to semaglutide?
- What prescribing information, specific to semaglutide and weight loss, could you share with your patient regarding realistic weight loss targets?
- In addition to teaching your patient proper injection technique for the use of semaglutide, what other lifestyle education behaviors should you discuss at this point?
- What information should you share with your patient regarding the long-term use of semaglutide and the potential risks of stopping this medication (as it relates to weight regain)?
Your patient decides to go ahead with the semaglutide regimen.
- What are some patient education guidelines regarding common side effects of this medication?
- How often is the dose increased? What is the maximum dose this patient can receive weekly?
Your patient wants to know how long she will be taking this medication.
- What talking points will you cover regarding the long-term use of this medication?
- How do you best prepare this patient for long-term success with this medication?
- What lifestyle behavior modification education would you discuss with your patient, to give her the best chance at successfully managing her diabetes?
References + Disclaimer
Safe Prescribing of Opioids
- Bhatnagar, P. (2022, Deptember 12). Opioid Equivalency. StatPearls. Retrieved from https://www.ncbi.nlm.nih.gov/books/NBK535402/
- Center for Diesease and Control. (n.d.). Assessing benefits and harms of opioid therapy. Retrieved from CDC: https://www.cdc.gov/drugoverdose/pdf/assessing_benefits_harms_of_opioid_therapy-a.pdf
- Center for Disease Control. (2019, August 13). Prescribing Practices. Retrieved from CDC: https://www.cdc.gov/drugoverdose/deaths/prescription/practices.html
- Center for Disease Control. (2021, May 19). PDMPS: What States Need to Know. Retrieved from CDC Drug Overdose: https://www.cdc.gov/drugoverdose/pdmp/index.html
- Center for Disease Control. (2023, April 28). CDC’s 2022 Clinical Practice Guideline for Prescribing Opioids for Pain. (C. Mikosz, & M. Dowling, Eds.) Retrieved from CDC Training for Healthcare Professionals: https://www.cdc.gov/opioids/healthcare-professionals/training/overview.html
- Center for Medicare Advocacy. (2023, June 29). Medicare Enrollment Numbers. Retrieved from Center for Medicare Advocacy (2023): https://medicareadvocacy.org/medicare-enrollment-numbers/#:~:text=33%2C948%2C778%20are%20enrolled%20in%20Original,enrolled%20in%20Medicare%20Part%20D.
- Center for Medicare and Medicaid. (2023). EPCS Getting Started Quick Reference Guide. Retrieved from Center for Medicare and Medicaid: https://www.cms.gov/files/document/epcs-program-getting-started-quick-reference-guide.pdf
- Centers for Medicare & Medicaid Services. (2020, August 4). Medicare Program: Electronic Prescribing of Controlled Substances; Request for Information. Retrieved from Federal Register: https://www.federalregister.gov/documents/2020/08/04/2020-16897/medicare-program-electronic-prescribing-of-controlled-substances-request-for-information-rfi
- Diversion Control Division. (n.d.). Electronic Prescriptions for Controlled Substances (EPCS). Retrieved from Diversion Control Division: https://www.deadiversion.usdoj.gov/ecomm/e_rx/
- Diverson Control Division. (2023). Controlled Substance Schedules. Retrieved from US Departement of Justice Drug Enforcement Administration Diversion Control Division: https://www.deadiversion.usdoj.gov/schedules/index.html.
- Dowell, D., Ragan, K., Jones, C., & Baldwin, G. (2022, December 1). Prescribing Opioids for Pain — The New CDC Clinical Practice Guideline. The New England Journal of Medicine, 2011-2013. doi:10.1056/NEJMp2211040
- Dowell, D., Ragan, K., Jones, C., Baldwin, G., & Chou, R. (2022, November 3). CDC Clinical Practice Guideline for Prescribing Opioids for Pain. 71(No. RR-3), 1-95. United States: MMWR Recomm Rep. doi: http://dx.doi.org/10.15585/mmwr.rr7103a1
- DrFirst. (n.d.). State Mandates Driving EPCS and PDMP Utilization. Retrieved from DrFirst: https://drfirst.com/resources/regulatory-mandates/
- Food and Drug Administration. (2020, October 1). Safe Disposal of Medications. Retrieved from FDA: https://www.fda.gov/drugs/safe-disposal-medicines/disposal-unused-medicines-what-you-should-know
- Food and Drug Administration. (2023, August 9). Timeline of Selected FDA Activities and Significant Events Addressing Substance Use and Overdose Prevention. Retrieved from FDA: https://www.fda.gov/drugs/information-drug-class/timeline-selected-fda-activities-and-significant-events-addressing-substance-use-and-overdose
- Geyer, D. H. (2023, January 17). A brief history of morphine use. Retrieved from Mayo Clinic Press: https://mcpress.mayoclinic.org/opioids/history-of-morphine/#:~:text=1803%3A%20Morphine%20is%20discovered&text=After%20conducting%20several%20years%20of,because%20it%20made%20people%20sleepy.
- Li, K., Harris, K., Hadi, S., & Chow, E. (2007). What should be the optimal cut points for mild, moderate, and severe pain? Journal of Palliative Medicine, 10(6), 1338-1346. doi: 10.1089/jpm.2007.0087
- National Institute on Drug Abuse. (2023, August 7). Only 1 in 5 US adults with opioid use disorder received medications to treat it in 2021. Retrieved from National Institutes of Health: https://nida.nih.gov/news-events/news-releases/2023/08/only-1-in-5-us-adults-with-opioid-use-disorder-received-medications-to-treat-it-in-2021#:~:text=News%20Release-,Only%201%20in%205%20U.S.%20adults%20with%20opioid%20use%20disorder,to%20treat%20it%20in%
- PDMP TTAC. (2020, July 10). Mandatory Enrollment and Use of PDMPs. Retrieved from Prescrption Drug Monitoring Program Training and Technical Assistance Center: https://www.pdmpassist.org/pdf/TAG_Mandatory_Enrollment_Use_20200710.pdf
- SAMHSA. (n.d.). Talking with Your Teen About Opioids. Retrieved from Substance Abuse and Mendal Health Services Administration: https://www.samhsa.gov/sites/default/files/TTHY-Opioid-Broch-2020.pdf
- Substance Abuse and Mental Health Services Administration. (2023, June 9). Naltrexone. Retrieved from SAMHSA: https://www.samhsa.gov/medications-substance-use-disorders/medications-counseling-related-conditions/naltrexone
- Substance Abuse and Mental Health Services Administration. (2021). Medications for Opioid Use Disorder TIP 63. Retrieved from SAMHSA: https://store.samhsa.gov/sites/default/files/pep21-02-01-002.pdf
- Substance Abuse and Mental Health Services Administration. (2023, June 20). Methadone. Retrieved from SAMHSA: https://www.samhsa.gov/medications-substance-use-disorders/medications-counseling-related-conditions/methadone
- Substance Abuse and Mental Health Services Administration. (2023, June 7). Waiver Elination (MAT Act). Retrieved from SAMHSA: https://www.samhsa.gov/medications-substance-use-disorders/waiver-elimination-mat-act
- The Center for Disease Control and Prevention. (2021, January 26). Opioid Commonly Used Terms. Retrieved from CDC: https://www.cdc.gov/opioids/basics/terms.html
- UAMS Psychatric Research Institute. (2023). What is Buprenorphine. Retrieved from UAMS Psychiatric Research Institute: https://psychiatry.uams.edu/clinical- care/cast/buprenorphine/
- Zhang, K. (2021, June 7). Overview of the Opioid NDC and MME Analytical File Compiled by CDC. 1-37. United States: Division of Overdose Prevention, CDC.
Controlled Substances
- Abi-Aad, K., & Derain, A. (2023, August 17). Hydromorphone. Retrieved from Stat Pearls: https://www.ncbi.nlm.nih.gov/books/NBK470393/
- Adler, J., Ballantyne, J., Chou, R., Davies, P., Fanciullo, G., & Fine, P. (2009). Clinical Guidelines for the Use of Chronic Opioid Therapy in Chronic NonCancer Pain. The Journal of Pain, 113-130.
- Ahmad, F., Rossen, L., Sutton, P., & & Talih, M. (2018). Provisional drug overdose. National Vital Statistics Reports, pp. 1-12.
- Altman, R., Clark, M., Huddart, R., & Klein, T. (2018, October 28). Pharm GKB Summary: Oxycodone Pathway, Pharmacokinetics. Retrieved from Pharmacogenet Genomics: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6602093/#:~:text=Approximately%2072%25%20of%20an%20oxycodone,oxycodone%20is%20unconjugated%20%5B70%5D
- Atrux-Tallau, N., naimi, Z., & Jaudinot, E. (2022, November 4). Pharmocokinetics of Morphine Sulfate Orodispersible tablets and Bioequivalence with Immediate-Release Oral Morphine Sulfate Formulations in Healthy Adult SubjectsUnder fastin Conditions. Retrieved from Clinical Drig Investigation: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9705466/
- Baldwin, G., Compton, W. M., Han, B., Houry, D., Jones, C. M., & Vivolo-Kantor, A. (2021, September 24). Methamphetamine use in the United States: epidemiological update and implocations for prevention, treatment and harm reduction. Annual New York Academy of Science. New York: National Institute of Health.
- Berg, K. A., & Clarke, W. P. (2018). Making Sense of Pharmacology: Inverse Agonism and Functional Selectivtiy. International Journal of Neuropsychopharmacology, 962-977.
- Bistas, K., Lopez-Ojeda, W., & Ramos-Matos, C. (2023, May 29). Fentanyl. Retrieved from Stat Pearls: https://www.ncbi.nlm.nih.gov/books/NBK459275/
- Blyth, F., Briggs, A., Hoy, D. G., March, L. M., & Schneider, C. H. (2019). The Global Burden of Musculoskeletal Pain-Where to From Here? American Journal of Public Health, 35-40.
- Bramanti, P., Cavalli, E., Mammana, S., Mazzon, E., & Nicoletti, F. (2019). The neuropathic pain: An Overview of the current treatment and future therapeutic approaches. International Journal of Immunopathology and Pharmacology, 33.
- Brennan, F., Carr, D., Cousins, M., & Pain, D. (2013). Pain Management: A Fundamental Human Right. Anesthesia and Analgesia, 224-227.
- Carver, A. C., & Foley, K. (2003). Types of Pain. In D. Kufke, & R. W. Pollock, Cancer Medicine. Hamilton: Holland Frei Cancer Medicine.
- Centers for Disease Control and Prevention. (2022). CDCs Clinical Practice Guidelines for Prescribing Opioids. Retrieved from Opioids: https://www.cdc.gov/opioids/healthcare-professionals/prescribing/guideline/index.html?s_cid=DOP_Clinician_Search_Paid_001&gclid=CjwKCAjwrranBhAEEiwAzbhNtVKOCMzLLnwhfj7B2D_aBCZM_SryNcEyXl0GRigmorFhKve2ekCVrhoCNYQQAvD_BwE
- Centers for Disease Control and Prevention. (2023, August 8). Opioids. Retrieved from Centers for Disease Control and Prevention: https://www.cdc.gov/opioids/basics/epidemic.html#print
- Chiang, N., Gyaw, S., & Krieter, P. A. (2019). Comparison of the Pharmacokinetic Properties of Naloxone Following the Use of FDA-Approved Intranasal and Intramuscular Devices Versus a Common Improvised Nasal Naloxone Device. Journal of Clinical Pharmacology, 1078-1084.
- Cicero, T., & Ellis, M. &. (2017). The changing face of heroin use in the United States: A retrospective analysis of the past 50 years. JAMA, 673-681.
- Cofano, S., & Yellon, R. (2022, October 24). Stat Pearls: Hydrocodone. Retrieved from National Institutes of Health: https://www.ncbi.nlm.nih.gov/books/NBK537288/
- Dart, R., Hoyte, C., & Nakhee, S. (2021). A Review on Tramadol Toxicity: Mechanism of Action, Clinical Presentation, and Treatment. Forensic Toxicology, 293-310. Retrieved from Forensic Toxicology: https://link.springer.com/article/10.1007/s11419-020-00569-0
- Delsignor, R. e. (2001). Lofexidine versus clonidine in rapid opiate detoxification. Journal of Substance Abuse Treatment, 11-17.
- Department of Defense Veterans Association. (2023). Defense and Veterans Pain Rating Scale. Retrieved from VA.gov: https://www.va.gov/PAINMANAGEMENT/docs/DVPRS_2slides_and_references.pdf
- Department of Health and Aged Care. (2013, January 20). FLACC Pain Scale. Retrieved from health.gov: https://www1.health.gov.au/internet/publications/publishing.nsf/Content/triageqrg~triageqrg-pain~triageqrg-FLACC
- Donaldson, S. L. (2017). Provider-Based Interventions to Mitigate Risk for Opioid Pain Medication Abuse Among Adult Patients in a Primary Care Setting.
- Dydyk, A. M., & Grandhe, S. (2023, January 29). Pain Assessment. Retrieved from STAT PEARLS: https://www.ncbi.nlm.nih.gov/books/NBK556098/#:~:text=Pain%20is%20the%20most%20common,pain%20in%20the%20United%20States.
- Fan, M., Hamilton, M., Hyland, B., & Tscheng, D. (2019). Diversion of Controlled Drugs in Hospitals: A Scoping review of Contributors and Safeguards. Journal of Hospital Medicine.
- GeriatricPain.org. (2022, January). Fast Facts: Selected Pain Assessment Tools. Retrieved from Geriatricpain.org: Geriatricpain.org https://geriatricpain.org/selected-pain-assessment-tools-fc
- Haffajee, R., Jena, A., Weiner, S., & & Kesselheim, A. (2019). Mandatory use of prescription drug monitoring programs. Journal of the American Medical Association, 1667-1668.
- International Association for the Study of Pain . (2020, July 16). IASP Announces Revised Definition of Pain. Retrieved from International Association for the Study of Pain: https://www.iasp-pain.org/publications/iasp-news/iasp-announces-revised-definition-of-pain/
- JAMA. (2023). Substance Use and Addiction Medicine. Retrieved from JAMA Network: https://jamanetwork.com/collections/5921/substance-use-and-addiction-medicine
- John, W. S., Schwartz, R. P., Subramanian, G., & Wu, L.-T. (2018). Prevalence, Patterns and correlates of multiple substance use disorders among adults. Drug and Alcohol Dependency, 79-87.
- Jones, C., Einstein, E., & Compton, W. (2018). Changes in synthetic opioid involvement in drug overdose deaths in the United States. JAMA, 1819-1821.
- Kalava, A., King, K. C., & Preuss, C. V. (2023, April 29). Perscription of Controlled Substances: Benefits and Risks. Retrieved from STAT PEARLS: https://www.ncbi.nlm.nih.gov/books/NBK537318/
- Khalid, N., Patel, P., & Singh, A. (2023, January). Cannabis Versus Opioids for Pain. Retrieved from Stat Pearls: https://www.ncbi.nlm.nih.gov/books/NBK573080/
- Kreutzwiser, D., & Tawfic, Q. (2020). Mathadone for Pain Management. A Pharmacotherapeutic Review, 827-839.
- Kroenke, K., Spiter, R., Williams, J., & Lowe, B. (2011, Nov). An ultra-brief screening scale for anxiety and depression: the PHQ-4. From Principles of Neuropathic Pain Assessment and Management, 613-21. Psychosomatics 2009.
- Manchikanti, L., Kaye, A., Knezevic, N., McAnally, H., Slavin, K., Trescot, A., & Hirsch, J. (2018). Responsible, safe, and effective prescription of opioids for chronic non-cancer pain: American Society of Interventional Pain Physicians guidelines. Pain Physician, S3-S92.
- Marquez, C. B. (2020). Changing the outlook toward the problem of opioid addiction: From “War on Drug” to “Public Health Emergency”. Seton Hall: Law Student Scholarship.
- Marsch, L., Borodovsky, J., & Dallery, J. (2019). Advances in the psychosocial treatment of addiction: The role of technology in the delivery of evidence-based psychosocial treatment. The Psyciatric Clinics of North America, 585-596.
- McLellan, T., & Volkow, N. (2016). Opioid Abuse in Chronic Pain-Misconceptions and Mitigation Strategies. New England Journal of Medicine, 1253-1263.
- National Institute of Drug Abuse (NIDA). (2023, August 30). Prescription Opioid Drug Facts. Retrieved from National Institute on Drug Abuse: https://nida.nih.gov/publications/drugfacts/prescription-opioids
- National Institute of Health. (2023, August 25). Medications for Opioid Overdose, Withdrawal, & Addiction. Retrieved from National Institute of Drug Addiction: https://nida.nih.gov/research-topics/opioids/medications-opioid-overdose-withdrawal-addiction-infographic
- Pain Physician. (2008). Opioid Special Issue. Pain Physician, S133-S153.
- Ransford, e. a. (2019, February 08). The Ransford Pain Drawing . Retrieved from compassionandsupport.org: https://compassionandsupport.org/wp-content/uploads/sites/2/2019/08/PainScaleRansford.pdf
- Rudd, R. A., Zibbell, J., & Gladden, M. (2016). Increases in drug and opioid overdose deaths-Untied States. Morbidity and Mortlity Weekly Report, 1378-1382.
- SAMHSA. (2020). Key Substance Use and Mental Health Indicators in the United States: Ressults from the 2019 National Survey on Drug Use and Health. SAMHSA.
- Scalpel, S. (2016, November). The Joint Commission Deserves Some Blame for the Opioid Crisis. Retrieved from Missouri Medicine: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6139759/
- Scholl, L., Seth, P., Kariisa, M., Wilson, N., & Baldwin, G. (2019). Drug and opioid-involved overdose deaths-Unioted States, 2013-2017. Morbidity and Mortatlity Weekly Report 67, 1419-1427.
- Stauffer, L., & Gibson, S. (2017). The Nurse’s Role in Identifying and Educating Patients At Risk for Opioid Abuse. AAACN Viewpoint, 1.
- The Joint Commission. (2023, January). The Joint Commission History Timeline. Retrieved from JointCommission.org: https://www.jointcommission.org/-/media/tjc/documents/tjc-history-timeline-through-2022.pdf
- Toombs, K., Kral, L., Colleran, C., & & Varney, S. (2018). Nurse practitioner-pharmacist collaboration in pain management: Strategies for success. . Journal of the American Association of Nurse Practitioners, 141-148.
- U.S. Food and Drug Administration. (2023, May 23). FDA. Retrieved from Information about Medication Assisted Treatment (MAT): https://www.fda.gov/drugs/information-drug-class/information-about-medication-assisted-treatment-mat
- Volkow, N., Jones, E., Einstein, E., & Wargo, E. (2019). Prevention and Treatment of Opioid mIsuse and Addiction: A Review. JAMA Psychiatry, 208-216.
Tirzepatide for Type 2 Diabetes and Weight Management
- Katie Kerwin McCrimmon, Uch. (2023, May 15). What is Mounjaro? and does it work better for weight loss than Ozempic and Wegovy?. UCHealth Today. https://www.uchealth.org/today/what-is-mounjaro-and-how-does-it-work-for-weight-loss/
- National Institutes of Health. (2023, July 28). DailyMed – mounjaro- tirzepatide injection, solution. U.S. National Library of Medicine. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=d2d7da5d-ad07-4228-955f-cf7e355c8cc0
- Type 2 Diabetes Treatment to Lower A1C | Mounjaro® (tirzepatide). (2023). https://www.mounjaro.com/?utm_id=go_cmp-16966473997_adg-133042365262_ad-626584063672_kwd-1651326559888_dev-c_ext-_prd-_mca-_sig-EAIaIQobChMIxcXZqYeHgQMVhDjUAR2Lvw5CEAAYASAAEgIMkfD_BwE&utm_source=google&utm_medium=ppc&campaign=16966473997&adgroup=133042365262&ad=626584063672&utm_keyword=kwd-1651326559888&gclid=EAIaIQobChMIxcXZqYeHgQMVhDjUAR2Lvw5CEAAYASAAEgIMkfD_BwE
- GoodRX – error. (2023, July 14). https://www.goodrx.com/mounjaro/how-make-you-feel
- Chen, S. (2023, July 28). Diabetes drug Mounjaro shown to have extraordinary weight loss for people without diabetes. diaTribe. https://diatribe.org/diabetes-drug-mounjaro-shown-have-extraordinary-weight-loss-people-without-diabetes
- VA Pharmacy Benefits Management Services. (2022, August). Tirzepatide Injection Criteria for Use. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Tirzepatide_MOUNJARO_Criteria_Aug_2022.pdf.
- Allied PR. (2023 B.C.E., May 6). Mounjaro Weight Loss: Newly Launch Over The Counter Alternative Pills To Tirzepatide & Mounjaro Weight Loss. https://newsdirect.com/news/mounjaro-weight-loss-newly-launch-over-the-counter-alternative-pills-to-tirzepatide-and-mounjaro-weight-loss-300908569. Retrieved August 30, 2023, from https://newsdirect.com/news/mounjaro-weight-loss-newly-launch-over-the-counter-alternative-pills-to-tirzepatide-and-mounjaro-weight-loss-300908569
- Mounjaro (2023). Savings and support for Mounjaro. Retrieved on August 31st 2023 from https://www.mounjaro.com/savings-resources?gclid=Cj0KCQjw9MCnBhCYARIsAB1WQVWAcINPKFwiHl4SbZQiGC8E_o9wHVOkDJDDUuphW3OlsKBagVTF1rQaApLJEALw_wcB
Semaglutide and Type 2 Diabetes
- Global diabetes cases to soar from 529 million to 1.3 billion by 2050. (n.d.). The Institute for Health Metrics and Evaluation. https://www.healthdata.org/news-events/newsroom/news-releases/global-diabetes-cases-soar-529-million-13-billion-2050.
- Diabetes Research Institute Foundation. (2023, March 31). Diabetes Statistics – DRIF. DRIF. https://diabetesresearch.org/diabetes-statistics/#:~:text=37.3%20million%20people%2C%20or%2011.3,yet%20been%20diagnosed%20(2022)
- Manage blood sugar. (2021, April 28). Centers for Disease Control and Prevention. https://www.cdc.gov/diabetes/managing/manage-blood-sugar.html
- About the National Diabetes Prevention Program | CDC. (n.d.). https://www.cdc.gov/diabetes/prevention/about.htm#:~:text=The%20National%20Diabetes%20Prevention%20Program%E2%80%94or%20National%20DPP%E2%80%94was%20created,diabetes%20in%20
- Ms, M. G. R. (2023, February 16). All about Ozempic. Healthline. https://www.healthline.com/health/drugs/ozempic#_noHeaderPrefixedContent
- Professional, C. C. M. (n.d.). Ghrelin. Cleveland Clinic. https://my.clevelandclinic.org/health/body/22804-ghrelin#:~:text=Ghrelin%20and%20leptin%20are%20two,brain%20when%20you’re%20hungry
- Prelipcean, M., MD. (2020, June 22). Behind the counter: Glucagon-like peptide-1 receptor agonists for type 2 diabetes. https://www.medicalnewstoday.com/articles/glp-1-receptor-agonists-type-2-diabetes
- Mba, A. B. P. (2023, May 1). Ozempic side effects: What you should know. https://www.medicalnewstoday.com/articles/drugs-ozempic-side-effects
- Delong, C. (2023, February 11). Black box warning. StatPearls – NCBI Bookshelf. https://www.ncbi.nlm.nih.gov/books/NBK538521/
- MNT Medical Network. (2023, February 16). Ozempic (semaglutide). https://www.medicalnewstoday.com/articles/326252
- Ozempic® vs. Other Type 2 Diabetes Treatments | Ozempic® (semaglutide) injection 0.5 mg or 1 mg. (n.d.). https://www.ozempic.com/why-ozempic/diabetes-medicines-comparison.html
- Northrop, A. (2023, August 3). Ozempic For Weight Loss: Side Effects, Risks And More. Forbes Health. https://www.forbes.com/health/body/ozempic-for-weight-loss/%20)
- Kerwin McCrimmon, UCHealth, K. (2023, April 5). Wegovy vs. Ozempic: The truth about new ‘weight-loss’ drugs. Uchealth. https://www.uchealth.org/today/wegovy-vs-ozempic-the-truth-about-new-weight-loss-drugs/#:~:text=If%20you%20take%20Wegovy%20or,to%20keep%20benefiting%20from%20them
- Stott, R. (2023, August 4). Lawsuit claims Ozempic, Mounjaro labels ‘downplayed severity’ of gastroparesis. Healio Gastroenterology. Retrieved August 11, 2023, from https://www.healio.com/news/gastroenterology/20230804/lawsuit-claims-ozempic-mounjaro-labels-downplayed-severity-of-gastroparesis#:~:text=While%20the%20drug%20label%20for,not%20recommended%20in%20these%20patients.%E2%80%9D
- How Does Gastroparesis Affect People with Diabetes? (2022, August 30). National Institute of Diabetes and Digestive and Kidney Diseases. https://www.niddk.nih.gov/health-information/professionals/diabetes-discoveries-practice/how-gastroparesis-affect-people-with-diabetes
- Living Healthy with Diabetes. (2023, March 29). www.heart.org. https://www.heart.org/en/health-topics/diabetes/prevention–treatment-of-diabetes/living-healthy-with-diabetes
- Move more; sit less. (2023, June 22). Centers for Disease Control and Prevention. https://www.cdc.gov/physicalactivity/basics/adults/index.htm#:~:text=Each%20week%20adults%20need%20150,Physical%20Activity%20Guidelines%20for%20Americans.&text=We%20know%20150%20minutes%20of,do%20it%20all%20at%20once
- Good sleep habits. (2022, September 13). Centers for Disease Control and Prevention. https://www.cdc.gov/sleep/about_sleep/sleep_hygiene.html
- Centers for Disease Control and Prevention. (2022, August 1). Food and nutrition insecurity and diabetes. Centers for Disease Control and Prevention. https://www.cdc.gov/diabetes/library/features/diabetes-and-food-insecurity.htm
Medication Assisted Treatment (MAT)
- American Psychiatric Association. (2020, December). What is a substance use disorder? https://www.psychiatry.org/patients-families/addiction-substance-use-disorders/what-is-a-substance-use-disorder
- Durrani M, Bansal K. Methadone. [Updated 2023 Apr 29]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK562216/
- Kumar R, Viswanath O, Saadabadi A. Buprenorphine. [Updated 2023 Apr 29]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK459126/
- National Center for Drug Abuse Statistics. (2023). Drug abuse statistics. https://drugabusestatistics.org/
- National Institute of Health. (2018, June). Understanding drug use and addiction. https://nida.nih.gov/publications/drugfacts/understanding-drug-use-addiction
- Singh D, Saadabadi A. Naltrexone. [Updated 2023 May 30]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK534811/
- Substance Abuse and Mental Health Services Administration. (2023, August). Methadone take-home flexibilities extension guidance. https://www.samhsa.gov/medications-substance-use-disorders/statutes-regulations-guidelines/methadone-guidance#:~:text=In%20treatment%200%2D14%20days,be%20provided%20to%20the%20patient
- S. Food and Drug Administration. (2023, May). Information about medication assisted treatment (MAT). https://www.fda.gov/drugs/information-drug-class/information-about-medication-assisted-treatment-mat
Disclaimer:
Use of Course Content. The courses provided by NCC are based on industry knowledge and input from professional nurses, experts, practitioners, and other individuals and institutions. The information presented in this course is intended solely for the use of healthcare professionals taking this course, for credit, from NCC. The information is designed to assist healthcare professionals, including nurses, in addressing issues associated with healthcare. The information provided in this course is general in nature and is not designed to address any specific situation. This publication in no way absolves facilities of their responsibility for the appropriate orientation of healthcare professionals. Hospitals or other organizations using this publication as a part of their own orientation processes should review the contents of this publication to ensure accuracy and compliance before using this publication. Knowledge, procedures or insight gained from the Student in the course of taking classes provided by NCC may be used at the Student’s discretion during their course of work or otherwise in a professional capacity. The Student understands and agrees that NCC shall not be held liable for any acts, errors, advice or omissions provided by the Student based on knowledge or advice acquired by NCC. The Student is solely responsible for his/her own actions, even if information and/or education was acquired from a NCC course pertaining to that action or actions. By clicking “complete” you are agreeing to these terms of use.
➂ Click Complete
To move on to the next lesson.
